Chapter 12: Crossing the Blood-Brain Barrier
Chapter 12: Crossing the Blood-Brain Barrier
Part IV: What Your Body Does With Coffee
The espresso was good that morning. I remember that specifically — a clean, bright shot from a bag of Ethiopian beans I’d been working through that week, pulled a few seconds short of where most people would stop it. I was at my desk by seven-thirty, and by seven-fifty, the world had sharpened. The screen looked crisper. My thoughts moved faster. The vague, fuzzy reluctance that accompanies early mornings had been replaced by something more alert, more directed. I could feel it happening, the way you can feel a tide coming in if you’re standing ankle-deep in the water.
Twenty minutes. That’s roughly how long it takes. You drink the coffee, the caffeine is absorbed through the lining of your stomach and small intestine, it enters your bloodstream, and then — this is the part that matters — it crosses a barrier that stops most molecules cold. It slips through the most selective checkpoint in your entire body, enters your brain, blocks the receptors that make you feel sleepy, and suddenly the morning makes sense.
I’ve had this experience thousands of times. Every coffee drinker has. But that morning was different, because the night before I had been staring at the results of our ADMET screening — the computational analysis that predicts how molecules are absorbed, distributed, metabolized, excreted, and how toxic they might be — and I had noticed something in the data that I hadn’t expected.
Caffeine crossing the blood-brain barrier was no surprise. That’s the whole reason coffee works as a stimulant. But the screen had flagged two other compounds as predicted to cross: cafestol and kahweol. The diterpenes. The same molecules from Chapter 2 that everyone associates with cholesterol — the ones that live in the oily fraction of unfiltered coffee, the ones that paper filters remove.
Diterpenes in the brain?
I set down the espresso and pulled the data up again. Checked the parameters. Ran the numbers a second time. The prediction held. Cafestol and kahweol had the right molecular properties to cross the blood-brain barrier. Not just marginally — they cleared the threshold comfortably.
I sat back in my chair and thought about all the hundreds of papers I’d read on coffee diterpenes over the years. Cholesterol metabolism. Bile acid regulation. FXR binding. Liver effects. That was the entire diterpene story as the field told it. Nobody — not a single paper I could recall — had ever seriously asked whether these molecules might reach the brain.
That surprised me. And in science, surprise is usually worth following.
The Most Selective Border in Your Body
To understand why the diterpene finding matters, you first need to understand what the blood-brain barrier is and why it exists.
Your brain is, in a sense, the most paranoid organ you own. It has reason to be. Neurons are exquisitely sensitive to chemical disruption. A slight shift in ion concentrations, an unwanted protein, a stray toxin — any of these can trigger seizures, cell death, or cascading dysfunction. The brain cannot afford to let just anything wander in from the bloodstream, which carries all manner of metabolic waste, dietary compounds, bacterial products, and environmental chemicals.
So the brain built a wall.
The blood-brain barrier, or BBB, is not a single structure you could point to on an anatomy chart. It’s a functional property of the blood vessels that supply the brain. In most of your body, the endothelial cells that line your capillaries have small gaps between them — fenestrations and loose junctions that allow molecules to pass relatively freely between the blood and surrounding tissues. This is how nutrients reach your muscles, how immune cells migrate to sites of infection, how drugs get to their targets in most organs.
But the capillaries in your brain are different. Their endothelial cells are locked together by structures called tight junctions — molecular rivets that seal the gaps between adjacent cells so thoroughly that almost nothing can squeeze through. If you imagine the walls of an ordinary capillary as a chain-link fence, the walls of a brain capillary are more like a welded steel plate. The fence lets things through; the plate doesn’t.
This means that to get into the brain from the bloodstream, a molecule essentially has two options. It can use a specialized transport system — a molecular escort that actively carries it across the barrier, the way a cargo ship carries goods through a locked canal. Glucose gets in this way, through dedicated glucose transporters, because the brain burns enormous amounts of energy and can’t survive without a constant glucose supply. Certain amino acids have their own transporters too.
Or the molecule can do what caffeine does: slip through on its own.
This second route — passive transcellular diffusion — is the one that matters for our story. To cross the BBB by passive diffusion, a molecule has to physically dissolve into the lipid membrane of the endothelial cell on one side, diffuse through the cell, and emerge on the other side into the brain. Think of it as swimming across a river of fat. To do that, the molecule needs certain properties. It needs to be small enough. It needs to be lipophilic enough — comfortable in a fatty environment. And critically, it can’t be too polar.
Polarity is the key concept here. Polar molecules have regions of uneven electrical charge — positive spots and negative spots — that make them attracted to water. Water is polar, which is why polar molecules dissolve well in it. But cell membranes are made of lipids, which are nonpolar. A molecule that’s too polar will be happily dissolved in the watery bloodstream but will refuse to enter the greasy interior of the endothelial cell membrane. It’s like trying to mix oil and vinegar — they don’t want to combine, and no amount of shaking will make them stay mixed for long.
So the blood-brain barrier is, fundamentally, a polarity filter. Small, nonpolar, lipophilic molecules slip through. Large, polar, hydrophilic molecules are turned away. And this simple physical principle — which has nothing to do with biological intelligence or active gatekeeping — explains why some drugs can reach the brain and others can’t, why alcohol affects your mind but sugar doesn’t make you drunk, and why pharmaceutical companies spend billions trying to redesign molecules to be just lipophilic enough to sneak past the barrier.
It also explains why I needed a way to measure polarity if I wanted to know which coffee compounds could reach the brain.
The TPSA Rule
In pharmaceutical research, the standard way to estimate whether a molecule can cross the blood-brain barrier is to calculate its topological polar surface area, or TPSA. This is exactly what it sounds like: a measurement of how much of the molecule’s surface is polar — occupied by oxygen atoms, nitrogen atoms, and the hydrogen atoms bonded to them. These are the parts of the molecule that “want” to interact with water and “don’t want” to pass through a lipid membrane.
TPSA is measured in square angstroms (Ų), where an angstrom is one ten-billionth of a meter — roughly the diameter of a single atom. It’s a molecular-scale ruler, measuring the area of the polar patches on a molecule’s surface the way you might measure the area of windows on the side of a building.
The empirical rule, established through decades of pharmaceutical data, is this: molecules with a TPSA below 90 Ų tend to cross the blood-brain barrier. Molecules above this threshold generally don’t. It’s not a law of physics — there are exceptions in both directions — but as a predictor, it works remarkably well across a wide range of drug candidates. Pharmaceutical companies use it routinely in the early stages of drug design, when they’re screening thousands of candidate molecules and need a quick way to estimate which ones have a chance of reaching the brain.
The beauty of TPSA is that it can be calculated directly from a molecule’s structure — you don’t need to run any experiments. You just need to know which atoms are present and how they’re connected. This made it ideal for our computational screening, where I was analyzing the molecular properties of coffee bioactives to predict their pharmacokinetic behavior.
There’s a second parameter that matters too: molecular weight. Large molecules — generally those above about 450 daltons — have a harder time crossing the BBB, even if their polarity is low. They’re simply too bulky to diffuse efficiently through the lipid membrane. So the combined rule of thumb is: TPSA below 90 Ų, and molecular weight below 450 Da. Meet both criteria, and you have a reasonable shot at reaching the brain.
These are empirical cutoffs, not absolute boundaries. I want to be clear about that. A molecule with a TPSA of 88 Ų isn’t fundamentally different from one at 92 Ų. The threshold is a statistical generalization based on observed patterns across hundreds of known drugs. But it’s a useful generalization, and when I applied it to our set of 15 key coffee bioactives, the results told a clear story.
Caffeine: The Positive Control
Let me start with the molecule nobody is surprised about.
Caffeine has a molecular weight of 194 Da — well below the 450 Da threshold. It’s a small molecule, compact and tidy. Its TPSA is low, meaning its surface is predominantly nonpolar. And it’s lipophilic enough to dissolve readily into cell membranes.
Everyone who has ever felt a coffee kick already knows, from direct personal experience, that caffeine crosses the blood-brain barrier. That alertness you feel twenty minutes after your first sip is the experiential proof. Caffeine enters your brain and blocks adenosine receptors — the receptors that accumulate a sleepiness signal throughout your waking hours. By blocking those receptors, caffeine doesn’t give you energy; it temporarily prevents your brain from receiving the message that you’re tired. The energy was already there. Caffeine just takes the brakes off.
For our ADMET screening, caffeine was important not because the result was surprising, but because it wasn’t. When your computational model predicts that caffeine crosses the BBB, and you know independently that caffeine absolutely does cross the BBB, that prediction serves as a positive control — a validation that the methodology is working correctly. If the model had predicted that caffeine couldn’t cross the barrier, I would have known something was wrong with the screening parameters.
It predicted correctly. Caffeine crossed. The model was behaving as expected.
Good. Now let’s talk about what it predicted for the molecules nobody expected.
The timeline of a coffee kick is a small masterclass in pharmacokinetics. When you take a sip of coffee, the liquid reaches your stomach within seconds. Caffeine — small, soluble, and stable in acidic conditions — begins to be absorbed through the stomach lining almost immediately, though most absorption happens a bit further along, in the upper part of the small intestine. Within about 15 to 20 minutes, caffeine concentrations in your bloodstream are rising sharply.
From the blood, caffeine reaches the blood-brain barrier. And here is where its molecular properties matter. At 194 Da, caffeine is small. Its polar surface area is low. It’s lipophilic. These three properties together mean it can dissolve directly into the lipid membranes of brain capillary endothelial cells and diffuse through them without needing any transporter or escort. It crosses the BBB by passive diffusion — the molecular equivalent of walking through an open door rather than needing a key.
Once in the brain, caffeine finds the adenosine A₂A receptors and sits in them, blocking adenosine from binding. Adenosine is a molecule that accumulates during waking hours and promotes drowsiness — it’s part of the body’s built-in sleep-pressure system. With caffeine occupying the receptors, the adenosine signal can’t get through, and you feel alert. The whole process — from sip to alertness — takes about 20 minutes. Not because any single step is slow, but because each step (gastric absorption, intestinal absorption, blood distribution, BBB crossing, receptor binding) takes a few minutes, and the minutes add up.
What’s remarkable is how perfectly caffeine’s molecular structure is suited to this journey. It’s as if the molecule were designed by a pharmaceutical company to be orally bioavailable, rapidly absorbed, and brain-penetrant. It wasn’t, of course. It evolved in the coffee plant as an insecticide. But the molecular properties that make it toxic to insects — its small size, its lipophilicity, its ability to cross biological membranes — are the same properties that make it the world’s most popular psychoactive drug.
The Surprise: Diterpenes Are Predicted to Cross
Now we get to the result that made me set down my espresso.
Cafestol, as I described in Chapter 2, has a molecular weight of 316.4 Da. Kahweol is slightly lighter at 314.4 Da. Both are well below the 450 Da cutoff. They’re diterpenes — molecules built from isoprene units arranged into a compact ring structure — and their overall architecture is lipophilic. They live in coffee oil, after all. They’re not water-loving molecules. They’re fat-loving molecules that dissolve in the lipid fraction of the brew and get caught by paper filters precisely because they associate with fats.
When I calculated their TPSA values, both cafestol and kahweol came in below the 90 Ų threshold. Their surfaces are predominantly nonpolar, with only a few polar patches contributed by the hydroxyl group and the furan ring oxygen. In terms of the two key parameters — polarity and size — they have the molecular profile of BBB-penetrant compounds.
Our ADMET screening flagged both of them as predicted to cross the blood-brain barrier.
I need to be very precise about what this means and what it doesn’t mean.
This is a computational prediction. It is based on the physical-chemical properties of the molecules — their size, their polarity, their lipophilicity — evaluated against empirical models derived from known drug data. The prediction says that cafestol and kahweol have the right molecular passport to pass through the blood-brain barrier by passive diffusion.
What the prediction does not tell us is whether cafestol or kahweol actually do cross the BBB in living humans. Nobody, to my knowledge, has ever measured cafestol concentrations in cerebrospinal fluid — the fluid that bathes the brain and spinal cord — after coffee consumption. Nobody has done the direct experimental measurement that would convert this prediction into an observation. The experiment hasn’t been done.
And the prediction certainly doesn’t tell us whether cafestol or kahweol do anything once they reach the brain — if they reach the brain. A molecule that crosses the BBB might be metabolized immediately. It might distribute to brain tissue at concentrations too low to have any biological effect. It might sit inertly in the lipid membranes without interacting with any receptor or enzyme. Crossing a barrier is not the same as having an effect on the other side.
I want to be explicit about this because I’ve seen too many computational predictions get inflated as they travel from the original paper into reviews, press releases, and popular articles. The chain typically goes: “predicted to cross” becomes “crosses” becomes “acts on the brain” becomes “protects the brain” becomes a headline about how your morning coffee is saving your neurons. Every step in that chain adds a claim that the data doesn’t support.
What I can say, with confidence, is this: the molecular properties of cafestol and kahweol strongly suggest they have the capacity to reach the brain. The computational prediction is robust — it’s the same methodology that correctly identifies caffeine as BBB-penetrant, and it’s based on well-established physical-chemical principles. This is a hypothesis worth testing experimentally. It is not yet an established fact.
But as a hypothesis, it’s a genuinely novel one. I spent weeks after running the ADMET screen going back through the diterpene literature — hundreds of papers stretching back to the 1980s — looking for anyone who had asked this question before. The entire field has been focused on the liver. Cholesterol metabolism. Bile acid regulation. FXR binding. The story of coffee diterpenes, as told by the scientific community for forty years, is a liver story.
Nobody, as far as I could find, had seriously considered that these molecules might be reaching the brain.
Figure 12.3. Longevity diets: how the world's longest-lived populations integrate coffee into dietary patterns associated with healthy aging.
Figure 12.4a. mTORC1 suppression: degree of mTOR pathway inhibition across different fasting protocols, from time-restricted eating to extended water fasts.
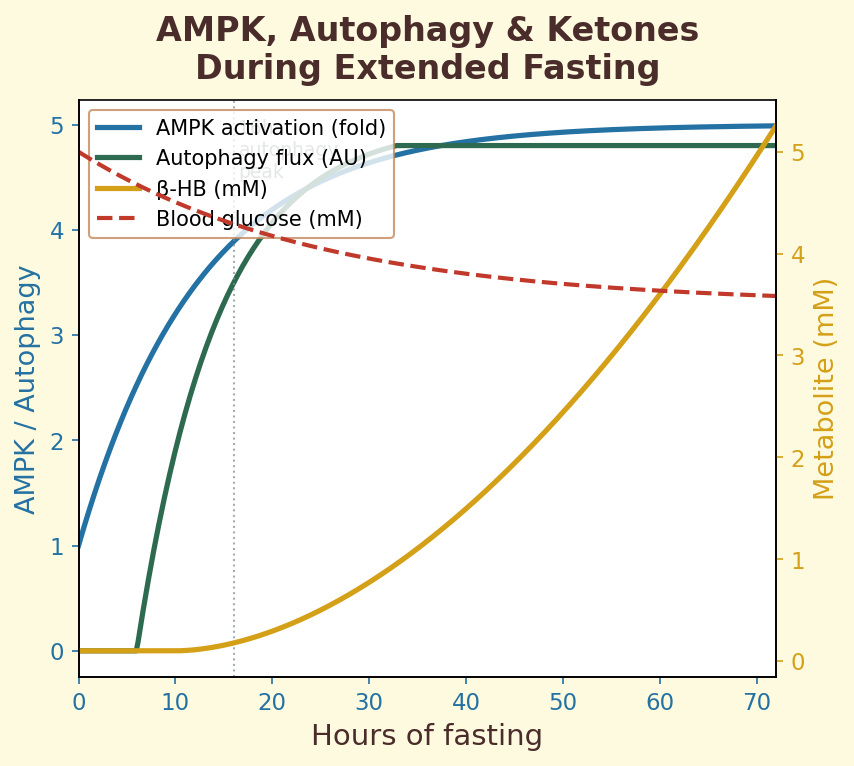
Figure 12.4b. AMPK activation and ketone production: reciprocal dynamics of the energy-sensing AMPK pathway and beta-hydroxybutyrate levels during progressive fasting.
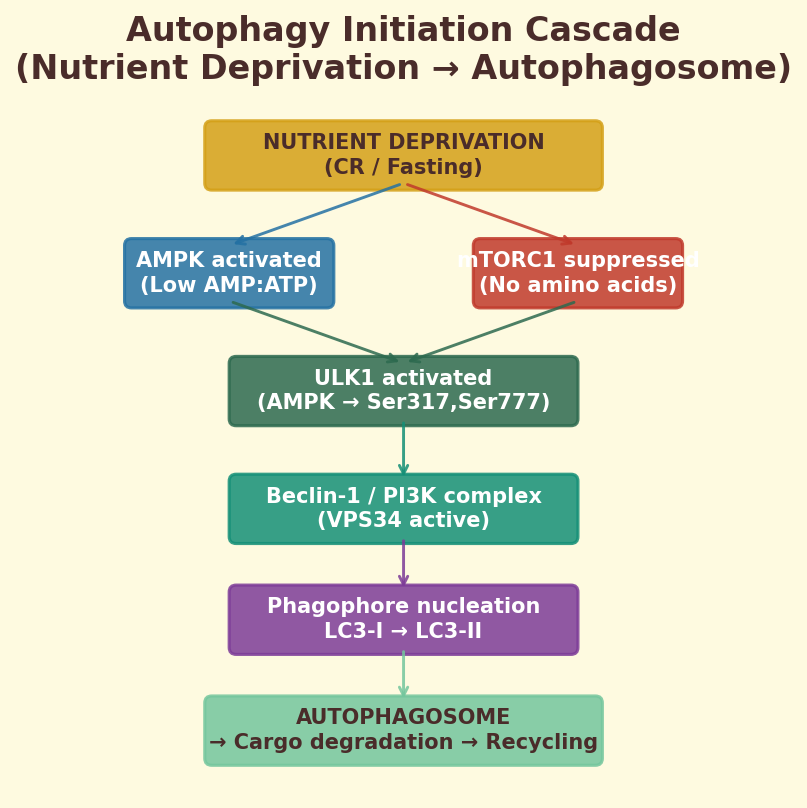
Figure 12.4c. ULK1 autophagy initiation: the molecular cascade from mTOR inhibition through ULK1 activation to autophagosome formation, with coffee compound intervention points annotated.
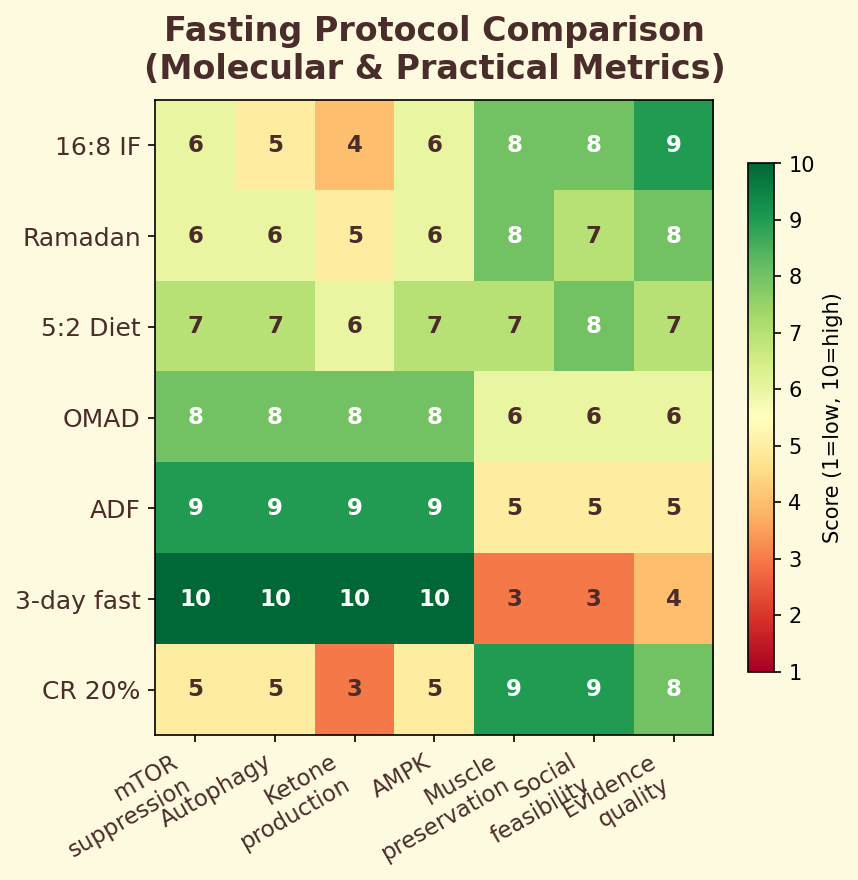
Figure 12.4d. Fasting protocol comparison: heatmap of metabolic, autophagic, and hormonal responses across intermittent fasting, alternate-day fasting, and extended fasting protocols.
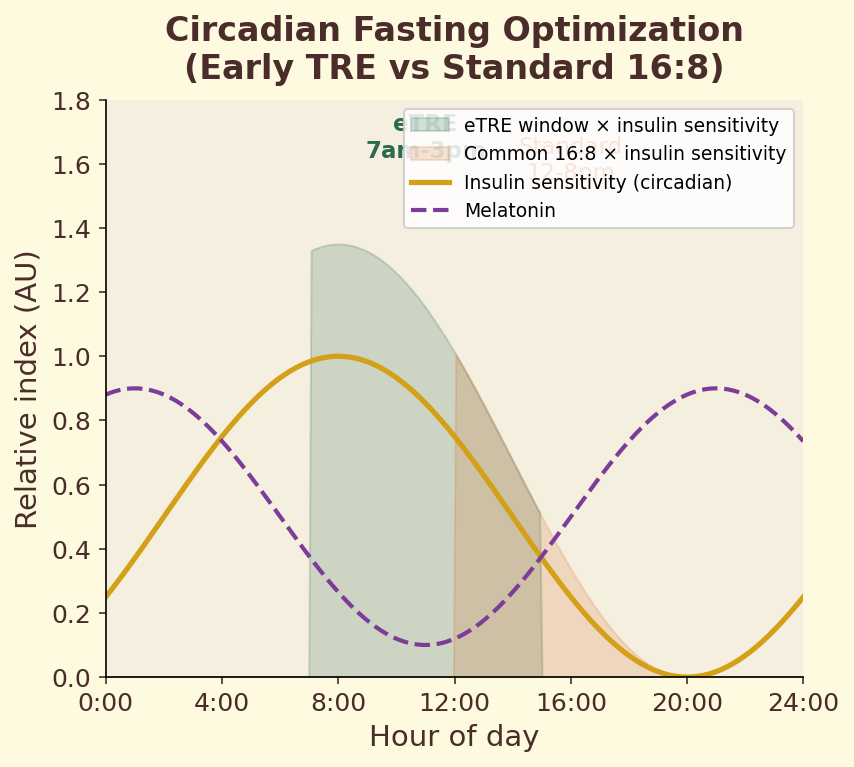
Figure 12.4e. Circadian fasting window: optimal timing of eating and fasting windows relative to circadian hormone rhythms, with coffee consumption timing overlay.
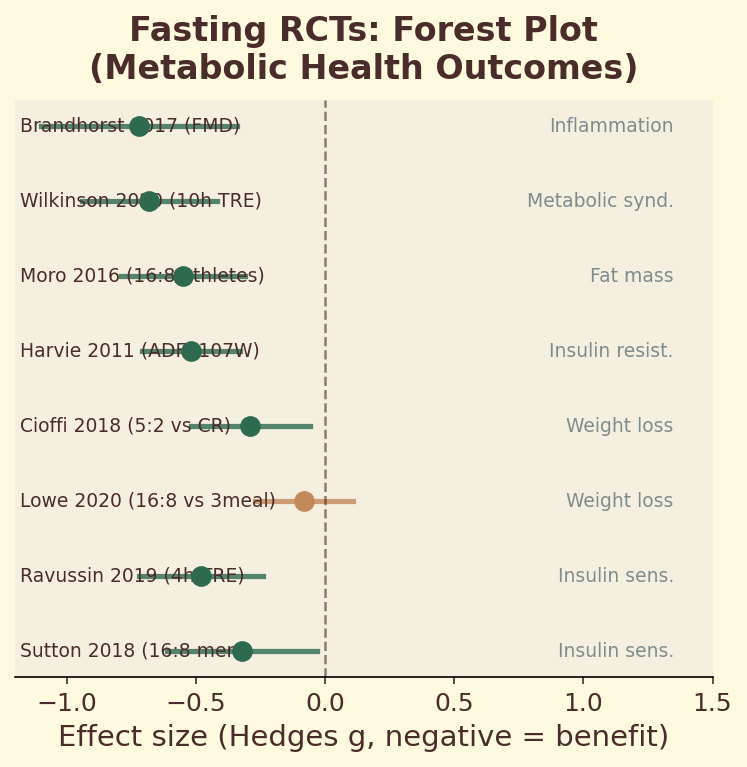
Figure 12.4f. Fasting RCTs: forest plot of randomized controlled trials measuring autophagy biomarkers in response to intermittent fasting interventions, with and without coffee consumption.
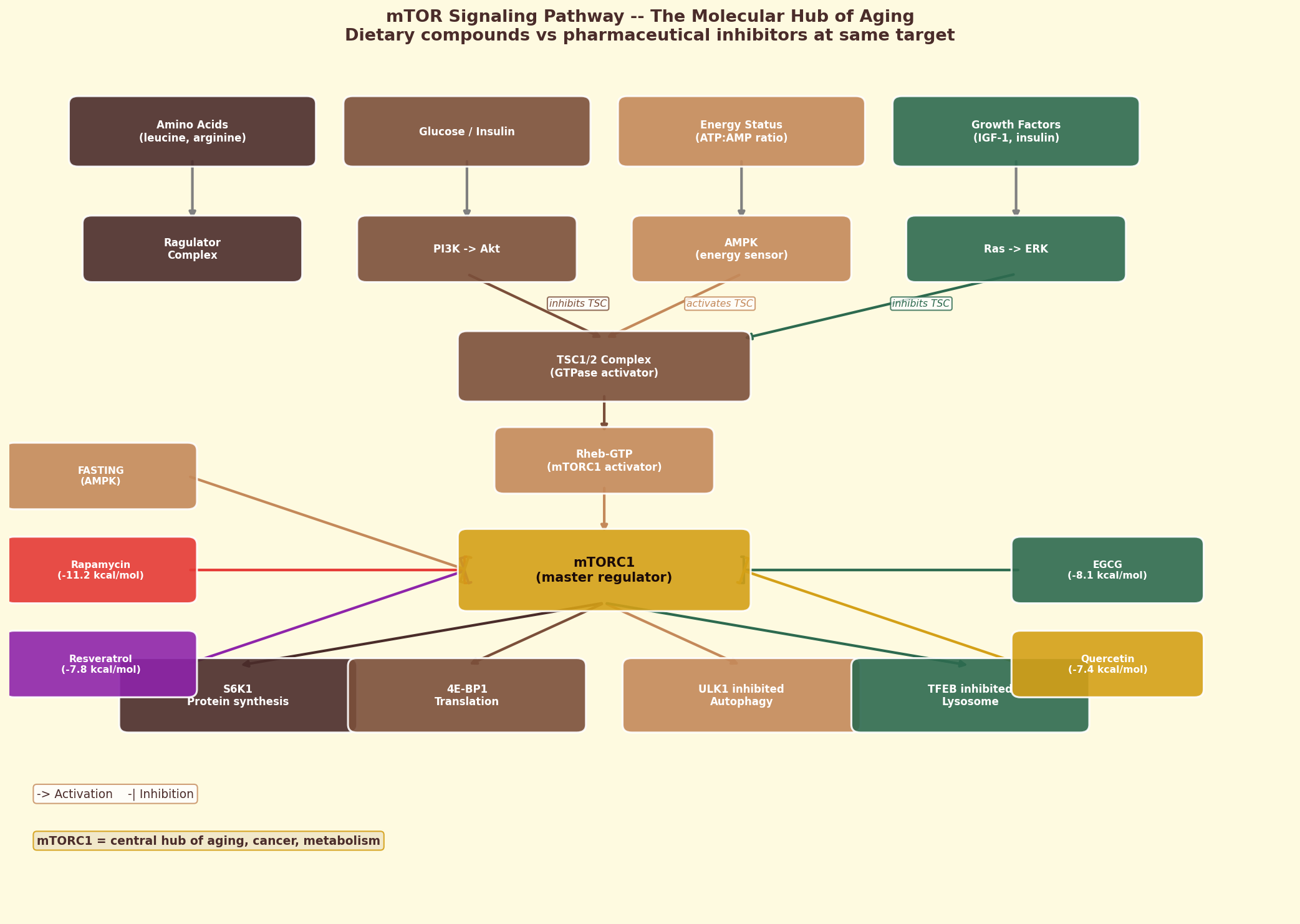
Figure 12.5. The mTOR signaling pathway: a master regulator of cell growth and metabolism, with multiple predicted interaction points for coffee bioactives.
Who Doesn’t Cross — And Why
The flip side of the prediction is equally important. Not every coffee bioactive is predicted to cross the BBB, and the molecules that are excluded tell us something significant about how coffee interacts with the body.
The most important exclusion is chlorogenic acids — the CGAs that are, by mass, the most abundant bioactive compounds in coffee. A single cup of coffee can contain tens of milligrams of chlorogenic acids, dwarfing the amount of any other bioactive present. They’re powerful antioxidants. They’ve been associated with a range of beneficial health outcomes in epidemiological studies. If you’ve read any popular science article about “the health benefits of coffee,” there’s a good chance chlorogenic acids were the star of the piece.
But their TPSA is 165 Ų.
That’s not just above the 90 Ų threshold — it’s nearly double it. I remember the moment that number appeared on my screen and I thought: well, there goes the obvious explanation. Chlorogenic acids are studded with hydroxyl groups, giving them a large, polar surface that makes them highly water-soluble and deeply incompatible with the lipid membranes of the blood-brain barrier. Our computational screening excludes them from BBB penetration, and the physical-chemical reasoning is unambiguous. These molecules are too polar to cross.
This matters enormously for how we interpret coffee’s associations with neurological outcomes. Large epidemiological studies have found associations between regular coffee consumption and reduced risk of conditions like Parkinson’s disease — a 28% risk reduction in meta-analyses. When researchers look for mechanisms to explain these associations, they naturally look at coffee’s most abundant bioactive compounds. Chlorogenic acids are the obvious candidate. They’re abundant, they’re biologically active, and they’re antioxidants.
But if chlorogenic acids can’t reach the brain, they probably aren’t responsible for neurological associations — at least not through direct brain-level mechanisms. The protective association, if it’s real and causal, would have to operate through some other pathway: peripheral effects that indirectly benefit the brain, gut-level mechanisms that influence brain health through the gut-brain axis, or — and this is where our prediction becomes intriguing — through other compounds that can reach the brain.
Compounds like caffeine. Compounds like caffeic acid and ferulic acid — the smaller phenolic acids that are released when chlorogenic acids break down during digestion, and which our screening predicts can cross the BBB because their individual molecular weights are lower and their TPSA values fall below the threshold. And compounds like cafestol and kahweol.
Here is one of the most interesting puzzles in coffee science, and one that rarely gets discussed in popular accounts.
Chlorogenic acids are coffee’s most abundant bioactive compounds. They’re among the strongest dietary antioxidants ever measured. They’ve been the subject of thousands of published studies, and they’re routinely invoked to explain coffee’s health associations — including neurological ones like reduced Parkinson’s risk.
But our computational screening predicts that CGAs can’t cross the blood-brain barrier. Their TPSA of 165 Ų — nearly double the 90 Ų threshold — makes them too polar to diffuse through the lipid membranes of brain capillaries.
So how do we reconcile these two facts? If CGAs can’t reach the brain, how could they contribute to neurological protection?
There are several possibilities, and they’re not mutually exclusive. First, CGAs are extensively metabolized during digestion. They’re broken down by gut bacteria and intestinal enzymes into smaller phenolic acids — caffeic acid, ferulic acid, and others — some of which are predicted to cross the BBB. So the neuroprotective compound might not be the CGA itself, but its metabolic children.
Second, CGAs are absorbed in the gut and are active right there — influencing inflammation, the leakiness of the gut lining, and which bacteria thrive. Your gut and your brain talk to each other constantly through what scientists call the gut-brain axis. This two-way communication system means that compounds acting in the intestine can influence brain health without ever physically entering the brain.
Third, CGAs may protect the brain indirectly through systemic effects — reducing whole-body inflammation, improving vascular health, regulating blood sugar. A healthier body provides a healthier environment for the brain.
The paradox reminds us that “reaching the brain” is not the only way a molecule can affect the brain. But it also means we should be cautious about assuming that coffee’s most abundant compound is necessarily its most neurologically relevant one.
The 10/15 Split
Let me step back and give you the full picture.
Our ADMET screening evaluated 15 key bioactive compounds in coffee — the molecules that are present at biologically relevant concentrations and have established or predicted biological activity. Of those 15, 10 are predicted to cross the blood-brain barrier.
The predicted crossers include caffeine — our positive control — along with both diterpenes (cafestol and kahweol) and several smaller phenolic acids, including caffeic acid and ferulic acid. These smaller phenolics are metabolic products of chlorogenic acid breakdown. When CGAs are digested, intestinal enzymes and gut bacteria cleave them into their component parts, releasing these smaller, less polar molecules. The parent CGA can’t cross the BBB, but the daughters can — at least according to our computational predictions.
The five compounds predicted not to cross are primarily the larger, more polar molecules — the chlorogenic acids themselves and other compounds with extensive hydroxyl group decorations that push their TPSA above the 90 Ų threshold.
What this means is that your morning coffee delivers a set of molecules to your bloodstream, and the blood-brain barrier acts as a filter — not unlike the paper filter in a drip coffee maker, though operating on entirely different physical principles. Some molecules pass through. Others are turned away. And the ones that pass through are not the ones that most people would guess.
If you asked a coffee chemist which of coffee’s compounds are most likely to reach the brain, they would say caffeine without hesitation, and then probably stop. If they added anything, it might be trigonelline. I doubt anyone would have said cafestol and kahweol. The diterpenes are liver molecules in the conventional telling. They’re the cholesterol story. They’re the reason you should or shouldn’t use a paper filter, depending on your cardiovascular risk factors.
But the TPSA data doesn’t care about conventional wisdom. It just measures polarity. And by that measure, the molecules everyone worries about for cholesterol have a molecular passport to the one organ everyone assumed they couldn’t reach.
What This Means — And What It Doesn’t
I’ve been a scientist long enough to know the distance between a computational prediction and a biological fact. It’s a long distance. Let me walk through exactly what we know, what we predict, and what we don’t know.
What we know: Caffeine crosses the blood-brain barrier. This is an established experimental fact confirmed by decades of pharmacological research and by the daily experience of billions of coffee drinkers.
What our computational screening predicts: Cafestol, kahweol, caffeic acid, ferulic acid, and several other coffee bioactives have the molecular properties consistent with BBB penetration. Their TPSA values are below 90 Ų, and their molecular weights are below 450 Da. By the standard pharmaceutical criteria used to evaluate drug candidates, these molecules are predicted to be brain-penetrant.
What we don’t know: Whether these molecules actually reach the brain in measurable concentrations after coffee consumption. Whether, if they do reach the brain, they interact with any biological targets there. Whether any such interaction is beneficial, harmful, or neutral. Whether the concentrations that could theoretically reach the brain are high enough to have any effect at all.
Predicting BBB penetration is not the same as predicting brain activity. A molecule that crosses the barrier might do nothing once it arrives. It might be metabolized by brain enzymes before it can interact with anything. It might distribute so diffusely that its concentration at any given receptor is negligible. Or it might do something we haven’t predicted — something that wouldn’t show up in a computational screen because we didn’t know to look for it.
These are computational predictions that generate hypotheses for future experimental work. The right next step — and I hope someone does this — would be to measure cafestol and kahweol concentrations in cerebrospinal fluid after controlled coffee consumption. That experiment would convert our prediction into an observation, or refute it. Either outcome would be valuable.
What I find most interesting about this result is not the prediction itself but the question it opens up. For forty years, the diterpene story has been a liver story — cholesterol, bile acids, FXR receptors, paper filters. That story is important, and it’s well-supported by evidence. But if cafestol and kahweol can reach the brain, then the diterpene story might be bigger than anyone thought. It might have a chapter that takes place above the neck.
And the CGA paradox — the fact that coffee’s most abundant bioactive compounds probably can’t reach the brain — reframes the neurological question entirely. If the epidemiological associations between coffee and reduced neurological disease risk are real and causal, the responsible molecules may not be the obvious ones. They may not be the most abundant ones. They may be the ones with the right molecular properties to slip past the most selective border in the body.
Here is what the blood-brain barrier chapter adds to the picture we’ve been building throughout this book.
Of the 15 key bioactive compounds in your coffee, 10 are predicted to cross the blood-brain barrier. The one everyone already knew about — caffeine — confirms that our computational methodology works. The novel ones — especially cafestol and kahweol, the diterpenes that the scientific literature has confined almost entirely to the liver story — open questions that nobody was asking before.
Meanwhile, the chlorogenic acids — coffee’s most abundant bioactive compounds, the ones most often credited with coffee’s health associations — are predicted not to cross. Their polar surface area is simply too large. If they contribute to neurological benefits, they probably do so through indirect pathways: gut-level effects, metabolic breakdown products, or systemic anti-inflammatory activity. Not by physically reaching the brain.
This doesn’t mean chlorogenic acids are unimportant. It means their importance might be operating through different mechanisms than we assumed. And it means the molecules that can reach the brain — caffeine, the diterpenes, the small phenolic acids — deserve more attention than they’ve received in the neurological conversation.
The next time you feel that familiar sharpening of focus twenty minutes after your first sip, you’ll know what’s happening at the molecular level. Caffeine has crossed the barrier, found the adenosine receptors, and blocked the sleepiness signal. That part of the story is well established.
But if our computational predictions are correct, caffeine may not be traveling alone. The diterpenes — especially if you’re drinking unfiltered coffee, French press, Turkish, or espresso — may be making the same crossing. What they do when they get there, if anything, is a question for the next generation of experiments.
Are diterpenes doing something in the brain? Nobody knows yet. But the computational prediction says they have the molecular passport to get there. And in science, getting to the right question is often more important than having the answer.
In the next chapter, we’ll follow the molecules that don’t cross the barrier — the ones that stay in the gut — and discover that staying behind doesn’t mean staying silent. The gut, it turns out, has its own ways of talking to the brain.
The counterintuitive NASA-validated trick where coffee + nap beats either alone
You'll Need
- One espresso or small strong coffee
- A timer
- A quiet place to nap
- Notebook
Do This
- Drink the espresso quickly (under 2 minutes).
- Immediately set a timer for 20 minutes and close your eyes.
- Don't worry if you don't fully fall asleep — even light rest works.
- When the timer goes off, rate your alertness immediately (1-10).
- Rate again at 30 min and 60 min after waking. Compare to your normal post-coffee alertness.
What's Happening
Caffeine takes 20-25 minutes to cross the blood-brain barrier and reach adenosine receptors. During a nap, your brain naturally clears adenosine (the sleepiness molecule). So you wake up with receptors freshly cleared AND caffeine arriving to block new adenosine from binding. NASA research showed this combination improved pilot alertness by 34% and performance by 16% compared to nap alone.
“All this chemistry depends on one practical choice you make every morning: how you brew.”