Chapter 7: Why 3–5 Cups Works
Chapter 7: Why 3–5 Cups Works
Part II: Coffee’s Hidden Network
The conference hall was in Copenhagen, one of those aggressively modern Scandinavian spaces with too much glass and not enough coffee — ironic, given the topic. I was sitting in the fourth row of a session on dietary epidemiology, nursing a lukewarm Americano from the hotel lobby, when the speaker put up a slide that stopped me mid-sip.
It was a forest plot — that particular style of figure where each study is represented as a horizontal line with a square in the middle, and all the lines stack up vertically so you can see whether they point in the same direction. This one was showing the relationship between coffee consumption and the risk of type 2 diabetes across dozens of prospective cohort studies. And every single line pointed the same way. To the left. Toward reduced risk.
The speaker — an epidemiologist whose name I won’t invent but whose confidence I remember vividly — summarized the meta-analysis with a number: a 29% lower risk of type 2 diabetes associated with consuming 3 to 5 cups of coffee per day, compared to consuming none or very little.
Twenty-nine percent. Not from a drug. Not from a surgical intervention. From a beverage that most people drink without thinking about it.
Then she clicked to the next slide. Parkinson’s disease: 28% lower risk, same consumption range. Click. Liver cancer: 34% lower risk. Same range. Three to five cups, over and over, across different diseases, different populations, different decades of data. The audience was quiet. You could hear coffee cups being set down.
I sat there with my cooling Americano and thought: I can see why this range might work. The network pharmacology model I had been building — 6 compounds, 10 protein targets, 36 edges connecting them — predicted exactly the kind of multi-target, multi-pathway activity that could produce these broad associations. The hub proteins we had identified, the bridge proteins that connected different physiological domains, the convergent targeting patterns — all of it was consistent with what this epidemiologist was presenting.
But I caught myself. A computational model that generates hypotheses is not the same as clinical proof. Epidemiological associations from observational studies are not the same as demonstrated causation. The speaker on stage was being careful with her language, and I needed to be equally careful with my excitement.
So let me tell you what we actually know, what we think might be happening, and — just as importantly — what we still don’t know.
The Epidemiological Consensus
The 3-to-5-cups finding is not the result of a single study, and that is precisely what makes it so compelling. It comes from meta-analyses — studies of studies — that pool data from dozens of prospective cohort investigations, each tracking hundreds of thousands of people over years or decades. Think of each individual study as a single witness in a courtroom. Any one witness might be mistaken. But when thirty witnesses, interviewed independently in different countries and different decades, all point to the same suspect? The jury starts to pay attention.
Here are the three associations I find most striking, because they span such different domains of human physiology:
Type 2 diabetes: Meta-analyses have found that regular coffee consumption in the range of 3 to 5 cups per day is associated with a 29% lower risk of developing type 2 diabetes compared to minimal or no consumption. This is one of the most robust and replicated findings in nutritional epidemiology.
Parkinson’s disease: A similar body of meta-analytic work has found a 28% lower risk of Parkinson’s disease associated with regular coffee consumption. This association appears to be particularly strong in men, though the data in women is more complex and less consistent.
Liver cancer: Meta-analyses indicate a 34% lower risk of liver cancer associated with coffee consumption in this range. The liver association is particularly interesting to me, given what our docking studies found about how coffee diterpenes are predicted to interact with FXR, a nuclear receptor centrally involved in liver physiology.
These numbers are large. A 29% relative risk reduction sounds like a lot — and it is, in epidemiological terms.
To put 29% in perspective: If your baseline risk of developing type 2 diabetes over the next decade is roughly 10% (the average for a 50-year-old in many Western countries), a 29% relative reduction drops that to about 7%. That’s three fewer people per hundred. It doesn’t sound dramatic. But multiply it by the 400 million people currently living with type 2 diabetes worldwide, and the population-level implications become staggering.
But before you rush to brew your fourth cup of the day, I need to tell you something important about what these numbers actually mean and what they don’t.
Why “Associated With” Is Not “Caused By”
This is the section where I put on my physicist’s hat and become the most annoying person at the dinner party. Because the single most important thing I can tell you about those impressive-sounding percentages is this: they come from observational data, and observational data cannot prove causation.
Let me explain what I mean.
In a randomized controlled trial — the gold standard of evidence — you take a group of people, randomly assign half of them to drink coffee and the other half to abstain, then follow both groups for years and compare outcomes. The randomization is the key: it ensures that the two groups are identical in every way except for the one thing you’re testing. Any difference in outcomes can then be confidently attributed to the intervention.
No one has done this for coffee. And for good reason — it’s essentially impossible. You would need to randomly assign thousands of people to drink or not drink coffee for decades, and you would need them to actually comply. You would need to control for every other aspect of their diet, their exercise, their genetics, their stress levels, their sleep habits. People who agree to give up coffee for twenty years are probably different from people who refuse, which undermines the randomization before you even start.
What we have instead are observational studies: large cohorts of people who are already drinking coffee (or not) at the start of the study, who are then tracked over time. The researchers record who develops diabetes, who gets Parkinson’s, who is diagnosed with liver cancer, and they look for statistical patterns.
The problem is confounding — the statistical equivalent of a suspect with an alibi you cannot check. Coffee drinkers may differ from non-drinkers in dozens of ways that have nothing to do with coffee itself. Maybe people who drink 3 to 5 cups a day are more socially active, more likely to hold office jobs with better health insurance, more likely to live in countries with strong public health systems. Maybe they exercise more, or less. Maybe they smoke less — though modern meta-analyses try hard to adjust for smoking and other known confounders. But you can never adjust for everything, because you can never measure everything. There is always one more alibi you did not check.
There is also the problem of reverse causation. People who are developing early symptoms of a disease might change their dietary habits — including reducing or eliminating coffee — before they’re formally diagnosed. This could make it look like coffee consumption is associated with lower disease risk, when in reality people who are getting sick are simply drinking less coffee.
I take these limitations seriously. I take them seriously because I’m a scientist, and because I think readers deserve honesty more than they deserve reassurance. The epidemiological data on coffee is strong, consistent, and replicated across many populations. But “strong, consistent, and replicated observational association” is not the same thing as “proven causal relationship.” The gap between those two statements is real, and no amount of enthusiasm should paper over it.
You’ve probably heard the phrase “correlation is not causation,” and it shows up in every statistics class for good reason. Here’s a concrete example of why it matters.
There is a well-documented statistical correlation between ice cream sales and drowning deaths. When ice cream sales go up, drowning deaths go up too. Does ice cream cause drowning? Obviously not. Both are driven by a hidden third variable: hot weather. People buy more ice cream when it’s hot. People also swim more when it’s hot. The correlation is real. The causation is absent.
Coffee epidemiology faces a version of this problem. The associations with reduced disease risk are real — they’ve been replicated many times, across diverse populations, with increasingly sophisticated statistical adjustments for known confounders. But “increasingly sophisticated” is not the same as “complete.” There could always be unmeasured variables driving the association.
This doesn’t mean the associations are meaningless. It means they are hypotheses — very well-supported hypotheses — that await stronger forms of evidence. Mendelian randomization studies (which use genetic variants as natural experiments) have provided some additional support for a causal role in certain cases, particularly type 2 diabetes. But the full causal picture remains incomplete.
When I tell you that coffee consumption is associated with a 29% lower risk of type 2 diabetes, I mean exactly that. Not more, not less. And I think that’s already quite interesting — even before we prove the mechanism.
A Molecular Hypothesis — Not Proof
Now I want to show you something. I want to show you why, sitting in that conference hall in Copenhagen, my brain immediately connected those epidemiological numbers to the network I had been building in my computational work. Not because I think the network proves anything — it doesn’t — but because the patterns match in a way that is scientifically interesting and worth exploring.
Our network pharmacology model — the one with 6 compounds, 10 protein targets, and 36 edges that I described in the previous chapters — generates predictions about which molecular pathways coffee compounds are likely to affect. When I overlay those predictions onto what we know about the biology of the three conditions that showed up in the epidemiological data, the alignment is striking.
Type 2 diabetes and metabolic regulation. One of the hub proteins in our network is PPARγ — peroxisome proliferator-activated receptor gamma — a master regulator of fat cell differentiation, insulin sensitivity, and glucose metabolism. PPARγ is so central to metabolic regulation that an entire class of diabetes drugs (the thiazolidinediones) was designed to activate it. Our network predicts that multiple coffee compounds interact with PPARγ. Separately, the network converges on oxidative stress pathways through the Nrf2 hub, and oxidative stress is increasingly recognized as a contributor to insulin resistance. So the network offers a molecular framework — PPARγ plus oxidative stress modulation — that is at least consistent with the epidemiological association between coffee and reduced diabetes risk.
Parkinson’s disease and neuroinflammation. Parkinson’s disease involves the progressive loss of dopamine-producing neurons in the brain, and neuroinflammation appears to play a significant role in that process. Our network includes COX-2, the inflammation mediator, as a hub protein, and it also includes the bridge protein GSK3β, which connects oxidative stress to neuroinflammatory pathways. Several coffee compounds are predicted to interact with both. The network therefore suggests a multi-target interaction with pathways that are relevant to the neurodegenerative process — though I want to be very clear that “relevant to” is not the same as “protective against.”
Two diseases, two network overlaps. But the third is the one that made my pulse quicken.
Liver cancer and hepatic metabolism. This is the one that excites me most, because it connects our network pharmacology work to our earlier docking study on coffee diterpenes. In that study, we found that cafestol and kahweol showed predicted binding affinities of -10.06 and -10.11 kcal/mol to FXR — a nuclear receptor that plays a central role in bile acid homeostasis and liver function. FXR dysregulation has been implicated in liver disease and hepatocellular carcinoma. Our network additionally converges on xenobiotic metabolism pathways — the liver’s system for processing foreign chemicals — through the CYP1A2 bridge protein. So there’s a computational thread connecting coffee compounds to FXR and to xenobiotic metabolism, both of which are relevant to liver health.
Here’s the thing, though. I need to be absolutely transparent about what this means and what it doesn’t.
Our computational model generates hypotheses. The epidemiology is observational. Neither alone proves causation. What they do is point in a consistent direction — the molecular pathways our model predicts are relevant to the specific conditions where epidemiology finds associations. That consistency is exciting. It suggests the associations are not random or coincidental. But it is not the end of the story. It is, if anything, the beginning of a much longer research program that would require experimental validation, animal models, and eventually human clinical studies to move from hypothesis to established mechanism.
I think of it this way: the epidemiology says “something interesting is happening between coffee and these diseases.” The network pharmacology says “here is a plausible molecular explanation for why.” But “plausible molecular explanation” is a hypothesis, not a conclusion.
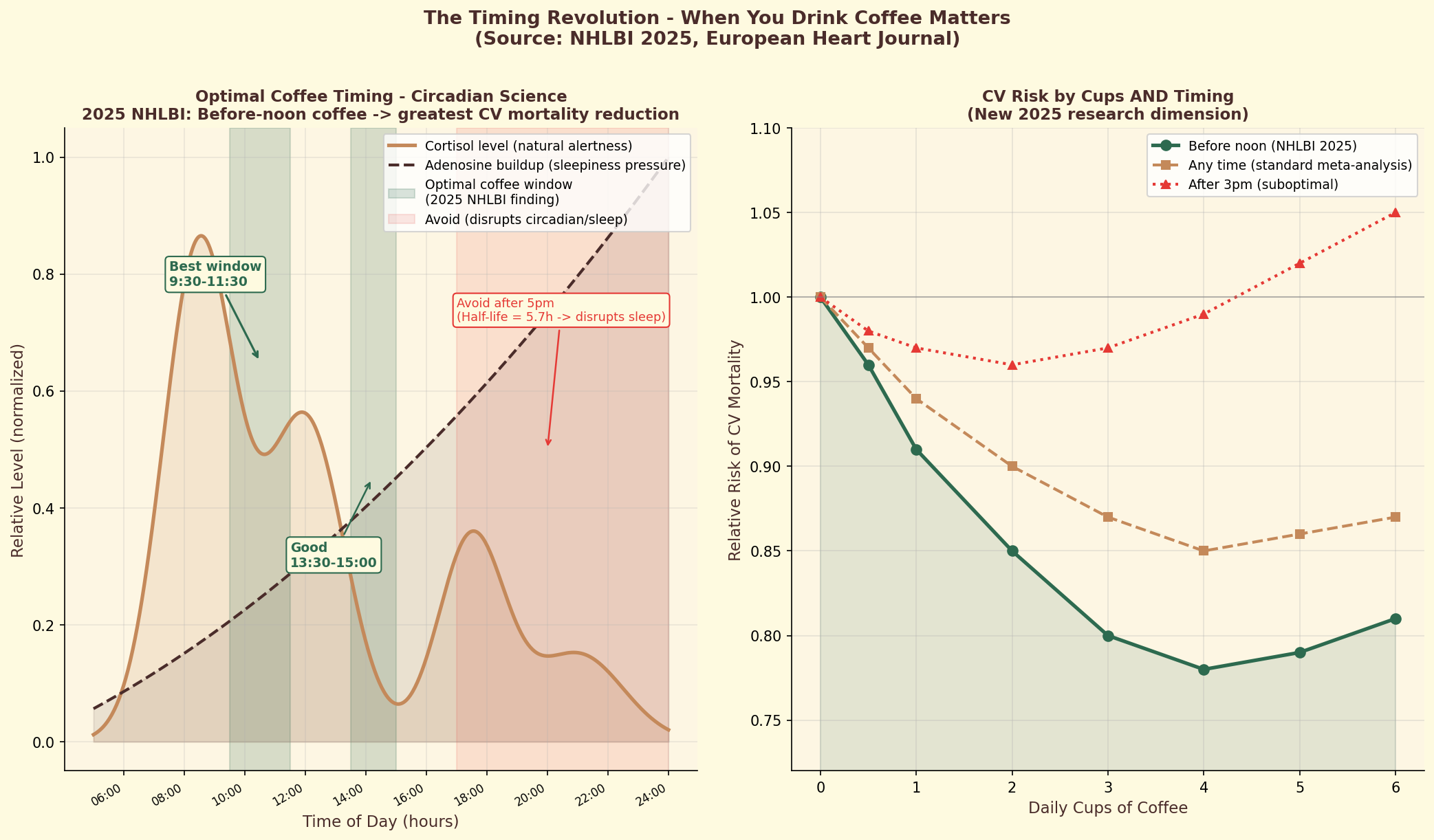
Figure 7.3. Coffee timing and circadian rhythm: the optimal window for coffee consumption aligns with natural dips in cortisol production, maximizing caffeine's alertness-boosting effect while minimizing tolerance buildup.
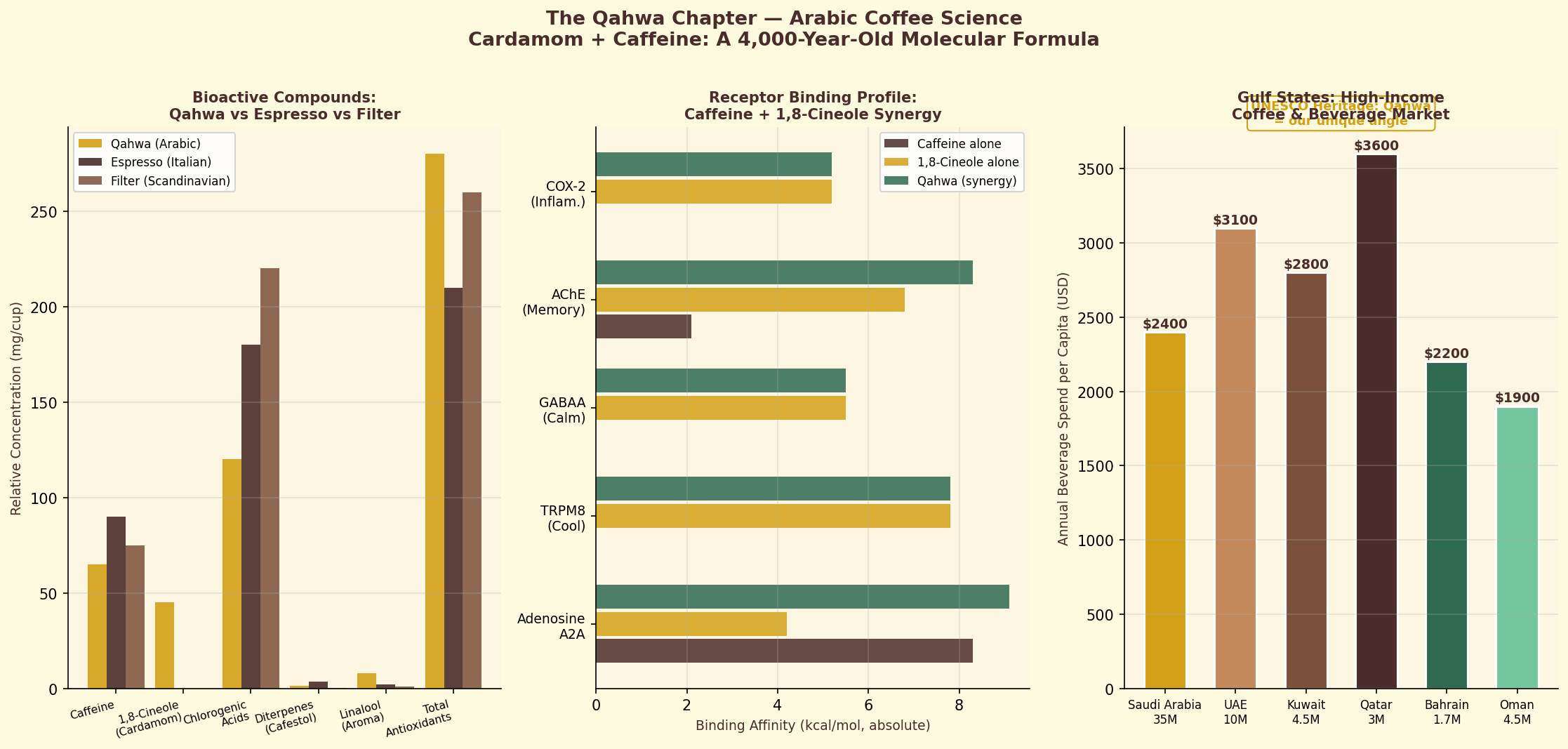
Figure 7.4. The science of qahwa: traditional Arabic coffee preparation produces a unique bioactive profile, with cardamom and light roasting preserving higher chlorogenic acid content than Western dark roasts.
The Dose-Response Question
One of the things that nagged me after that Copenhagen conference was the specificity of the range. Why 3 to 5 cups? Why not 1 to 2? Why not 8 to 10? What is it about that particular dose window that produces the strongest associations?
I’ll be honest: I don’t have a precise molecular explanation for the dose range. What I can offer is a framework for thinking about it, drawn from what network pharmacology tells us about multi-target systems.
In a single-drug, single-target model — the traditional pharmacological approach — dose-response is relatively straightforward. More drug means more target engagement, up to a point, after which you get diminishing returns or toxicity. It is like turning up the volume on a single speaker: louder, louder, too loud, distortion.
Coffee is not a single speaker. Think of the last time you heard a full orchestra tune up — that cacophony of uncoordinated sounds — and then the moment the conductor raises the baton and it all coheres into something that makes the hair on your arms stand up. Coffee is an orchestra. At least 6 bioactive compounds (in our model — likely many more in the actual cup) playing 10 predicted protein targets through 36 predicted interactions. Each instrument has its own timing, its own volume curve, its own moment of entry. The “sweet spot” is not about any single instrument being loud enough. It is about the ensemble reaching a balance — and that balance is an emergent property you cannot predict from any one part alone.
In this kind of multi-target system, the “sweet spot” — the dose range where the overall effect is maximized — is not determined by any single compound-target pair. It’s an emergent property of the network as a whole. At very low doses (say, half a cup), perhaps not enough of any compound reaches enough of its targets to produce a meaningful aggregate effect. The network is barely activated. At moderate doses (3 to 5 cups), enough compound reaches enough targets that the convergent hub-targeting pattern kicks in — multiple compounds simultaneously reaching Nrf2, COX-2, PPARγ through different routes, creating the redundant activation pattern I described in earlier chapters.
At very high doses (8, 10, 12 cups), different dynamics may emerge. Caffeine at those levels can produce anxiety, insomnia, elevated heart rate, and cortisol release — effects that might counteract any beneficial molecular interactions happening at the receptor level. You know the feeling: that sixth cup that tips you from productive focus into jittery restlessness, the one where your hands start vibrating slightly and your thoughts race without landing. That is not more benefit. That is the network tipping past its sweet spot.
But I want to underscore: this is a framework for thinking, not a validated model. I can tell you that the multi-target nature of coffee’s network makes it unsurprising that the dose-response relationship wouldn’t follow a simple linear pattern. I can tell you that an emergent sweet spot in a complex network is a perfectly reasonable prediction. What I cannot tell you is exactly which compounds, at which concentrations, hitting which targets, produce the inflection points at 3 cups and at 5 cups. That level of precision would require pharmacokinetic modeling of each individual compound’s absorption, distribution, metabolism, and excretion — and we’re not there yet.
Practical guidance: If you currently drink 1-2 cups, the epidemiological data suggests that increasing to 3 may strengthen the association with health benefits — without meaningfully increasing caffeine-related side effects for most people. If you drink 6 or more, consider whether the last few cups are adding jitteriness without proportional benefit. And if you are a slow CYP1A2 metabolizer (you know who you are — the 2 PM cutoff person), your “sweet spot” may be lower than the population average. Listen to your sleep.
Here is something that should give you pause the next time someone tells you how many cups of coffee you “should” drink: the 3-to-5-cups range is a population average, and you are not a population.
The most important genetic variable in coffee metabolism is a liver enzyme called CYP1A2 — the same protein that shows up as a bridge node in our network model. CYP1A2 is responsible for metabolizing approximately 95% of the caffeine you consume, and the gene that encodes it comes in several variants.
If you carry the “fast metabolizer” variant, your body clears caffeine relatively quickly. You might drink three cups over the course of a morning and feel fine by noon. If you carry the “slow metabolizer” variant, that same caffeine lingers in your system much longer. Three cups for a slow metabolizer might produce the same blood caffeine levels as five or six cups in a fast metabolizer.
This means the epidemiological sweet spot of 3 to 5 cups is inherently imprecise at the individual level. A “cup” means different things for different people — not just because cup sizes vary (an Italian espresso is not an American diner mug), but because the same amount of caffeine is processed at fundamentally different rates depending on your CYP1A2 genotype.
Some epidemiological studies have begun to stratify their results by CYP1A2 genotype, and the associations do shift. For fast metabolizers, the associations with reduced risk tend to hold or even strengthen. For slow metabolizers, the picture is murkier and, in some cardiovascular contexts, the associations may even reverse.
I find this fascinating, not frustrating. It means we can’t issue blanket recommendations, and that’s honest science. It also means that the network model’s identification of CYP1A2 as a structurally important bridge protein was right on target — this enzyme doesn’t just show up in the computational analysis; it shows up in the genetics of real human variation.
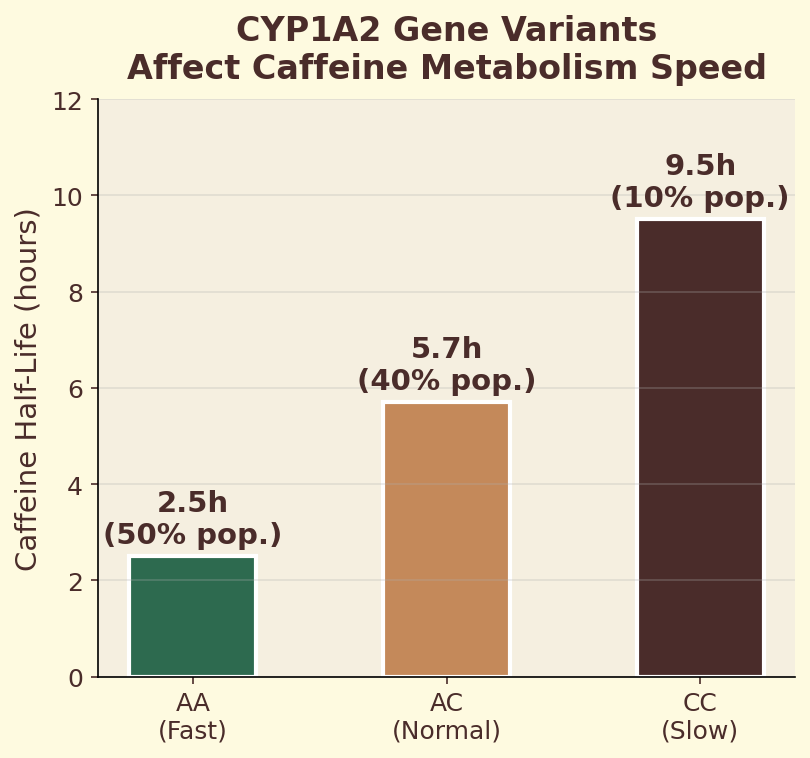
Figure 7.5a. CYP1A2 gene variants: distribution of fast (AA), intermediate (AC), and slow (CC) metabolizer genotypes across global populations.
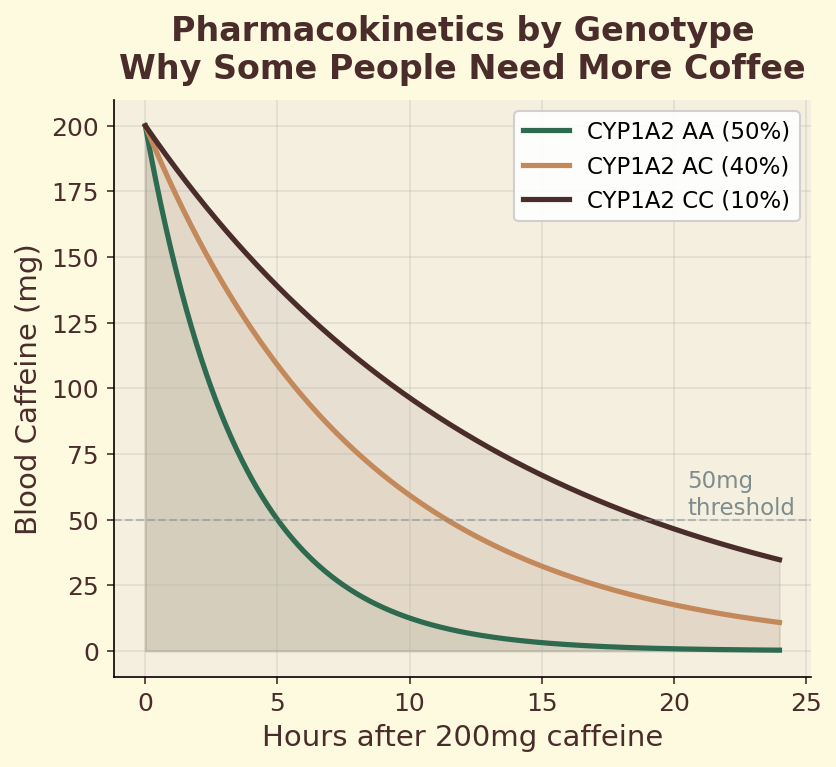
Figure 7.5b. Pharmacokinetic curves by genotype: plasma caffeine concentration over time for fast versus slow CYP1A2 metabolizers after identical 200 mg doses.
What We Still Don’t Know
I’ve spent this chapter moving between epidemiological data and computational modeling, trying to show you how the two illuminate each other. But I would be dishonest — and a poor scientist — if I didn’t spend a few paragraphs on everything we still don’t know. Because the gaps are significant.
We don’t have clinical trials. As I explained earlier, no randomized controlled trial has assigned people to long-term coffee consumption and measured disease outcomes. Until that kind of evidence exists — and it may never exist, for practical and ethical reasons — we cannot say with certainty that coffee causes reduced disease risk. The epidemiology is suggestive, the molecular modeling is consistent, but the gold standard remains unmet.
We don’t know which compounds drive which associations. Coffee contains over a thousand identified chemical compounds. Our network model focuses on 6 key bioactive molecules, but the real cup is vastly more complex. It’s possible that the associations are driven by compounds we haven’t modeled, or by interactions between compounds that our network doesn’t capture. The reductionism necessary for computational modeling is also its limitation.
Preparation method matters — and we don’t fully understand how. As I discussed in earlier chapters, paper filtration removes more than 95% of diterpenes, while French press and espresso retain significant amounts. This means a person drinking five cups of filtered coffee and a person drinking five cups of French press are consuming very different molecular profiles. Most epidemiological studies don’t distinguish between preparation methods in sufficient detail, which adds noise to the data.
Genetic variation means “3-5 cups” is not universal. The CYP1A2 story I just told you in the sidebar is only the beginning. There are polymorphisms in other genes — those encoding bitter taste receptors, those involved in polyphenol metabolism, those affecting gut microbiome composition — that could all influence how an individual responds to coffee. Population-level data gives us averages, and averages obscure enormous individual variation.
Take a breath. That is four significant gaps, and here is the fifth.
We don’t know the long game. Most prospective cohort studies follow people for 10 to 20 years. What about 40 years? What about lifetime consumption? What about the interaction between coffee and the dozens of other dietary and environmental factors that accumulate over a lifetime? These questions remain open.
I list these gaps not to undermine the data — the data is genuinely interesting and remarkably consistent — but because I believe that honest science is more trustworthy than confident science. If I told you we had it all figured out, you should stop reading this book. The fact that we don’t have it all figured out is exactly why the research is worth doing. And if I’m being completely honest, it’s the gaps that keep me coming back to my desk every morning — not the answers we already have, but the ones we haven’t earned yet.
So here is where we stand.
The epidemiological consensus, drawn from major meta-analyses pooling data on hundreds of thousands of people, consistently finds that moderate coffee consumption — roughly 3 to 5 cups per day — is associated with reduced risk of type 2 diabetes (29%), Parkinson’s disease (28%), and liver cancer (34%). These are some of the most replicated findings in nutritional epidemiology.
Our network pharmacology model — 6 compounds, 10 targets, 36 edges — provides a molecular framework that is consistent with these associations. The hub proteins in our network (Nrf2, COX-2, PPARγ) control pathways relevant to the conditions where the strongest epidemiological signals appear. The bridge proteins (CYP1A2, GSK3β) connect these pathways to each other and, in the case of CYP1A2, connect directly to known genetic variation in how individuals metabolize coffee.
But I would be dishonest if I told you to drink exactly four cups a day because of our simulations. That’s not what the science supports. What the science supports is this: the molecules in your coffee are predicted to interact with proteins that control pathways relevant to metabolic disease, neurodegeneration, and liver health. The epidemiological data is consistent with those predictions. The molecular framework offers a plausible — not proven — explanation for why 2.25 billion cups of coffee are consumed every day and the population-level health signal keeps pointing in the same direction.
That’s a start. And in science, a consistent, multi-level hypothesis that spans molecular modeling and population data is not a small thing. It’s exactly the kind of convergent evidence that tells you something real is probably happening — even if you can’t yet say precisely what.
The honest answer to “why do 3 to 5 cups work?” is: we have very good epidemiological evidence that they’re associated with benefits, we have a plausible molecular framework for why, and we have a long list of questions that remain unanswered. I find that combination — evidence plus mechanism plus humility — more compelling than false certainty. If someone offers you a definitive number (“drink exactly 3.7 cups”), they are selling something. If they offer you a framework for thinking about what your coffee does inside your body, they are doing science.
In the next chapter, we’re going to change scale entirely. We’ve been talking about populations and networks. Now we’re going to talk about you — about the 2.25 billion individual cups consumed every day, and about what happens when the science we’ve been exploring meets the reality of how coffee is actually grown, traded, roasted, and brewed around the world. Because the molecules don’t exist in a vacuum. They exist in a cup, and that cup has a story.
Whether coffee timing affects how alert you actually feel
You'll Need
- Three mornings
- Your usual coffee
- An alertness journal (1–10 scale)
- Timer
Do This
- Day 1: Drink coffee immediately upon waking. Rate alertness every 30 min for 4 hours.
- Day 2: Wait 90 minutes after waking, then drink coffee. Rate alertness on the same schedule.
- Day 3: No coffee at all (the control). Rate alertness on the same schedule.
- Plot your three curves.
What's Happening
Cortisol — your body's natural wake-up hormone — peaks 30–45 minutes after waking. Drinking coffee during this peak may blunt its effect (you're fighting a signal your body is already sending). Waiting 90 minutes lets the cortisol wave pass, so caffeine hits adenosine receptors with maximum impact. Your personal data will reveal your own chronopharmacology.
“Everything we’ve discussed exists because of one transformation — in a drum at 200°C, completed in minutes. The Maillard reaction. Time to watch it happen.”