Chapter 10: The Roaster's Bottleneck
Chapter 10: The Roaster’s Bottleneck
Part III: The Roasting Transformation
His name was Dani, and he roasted coffee in a converted garage in Zaragoza with the kind of obsessive precision that made me suspect he had been an engineer in a past life. He had a vintage Probat with a drum he had modified himself, a thermocouple setup that would make an analytical chemist nod approvingly, and a logbook — an actual paper logbook — where he recorded every roast he had done for the past six years. Temperature curves, batch weights, ambient humidity, even the wind direction on days when the garage door was open.
I had come to watch him roast a washed Ethiopian Yirgacheffe, and for the first forty minutes I mostly stayed quiet, watching the beans tumble behind the sight glass, listening to Dani narrate his process with the focus of someone defusing a bomb.
“Okay, we’re approaching first crack,” he said, leaning toward the drum. “This is where it gets interesting. From here, it’s all about development time.”
He used that phrase — development time — the way a surgeon might say “the critical window.” It meant the phase after first crack, the last sixty to ninety seconds of roasting where, according to every specialty roaster I have ever met, the entire flavor profile of the coffee is decided. Too short, and you get grassy, sour, underdeveloped flavors. Too long, and the coffee goes flat, baked, lifeless. The window is agonizingly narrow.
“You can’t rush it,” Dani said, adjusting his gas valve by what appeared to be a millimeter. “If you try to push through development with high heat, you get chaos. The outside of the bean develops and the inside stays raw. If you drop the heat too much, you stall, and everything tastes like cardboard.”
I had heard versions of this from every roaster I had visited over the previous two years. It was folklore — not in the dismissive sense, but in the sense that it was knowledge passed from roaster to roaster, refined by intuition, validated by taste, but never, as far as I could find, explained at the molecular level.
So I asked him the question I had been waiting to ask.
“What if I told you there’s a molecular reason why you can’t rush that phase? What if one specific chemical step is 75 to 125 times slower than the one before it?”
Dani looked at me the way people look at you when you have just said something that might be either very interesting or completely unhinged.
“I’d say tell me more,” he said.
The Phase Every Roaster Respects
Before I explain what my calculations found, I need to set the stage for anyone who has not stood next to a coffee roaster during those final minutes.
Coffee roasting is a thermal transformation that turns a dense, green, grassy-smelling seed into the fragrant brown bean you grind every morning. The process lasts roughly eight to fourteen minutes, depending on the roast profile, and it unfolds in stages that roasters track with almost surgical attention.
The early phase is mostly about drying — the green bean loses moisture, turns from green to yellow, and begins to smell like toasted bread. Then comes the Maillard reaction, the sprawling cascade of chemical transformations between sugars and amino acids that produces hundreds of flavor compounds, browning, and the characteristic aroma of roasted coffee. As temperature continues to rise, pressure builds inside the bean — water vapor and carbon dioxide trapped within the cellulose matrix push outward until the structure physically fractures. That fracture is audible. Roasters call it first crack, and it sounds like popcorn: a sharp, snapping report that tells you the bean has crossed a threshold.
After first crack, you enter the development phase. And this is where every roaster I have spoken to becomes reverent. Development is the phase where flavor complexity is built, where the difference between a bright, sweet, complex cup and a flat, hollow one is decided. It typically lasts between sixty and ninety seconds — sometimes slightly less, sometimes slightly more — and the margin for error is thin.
Every roaster knows this empirically. They know it from thousands of batches, from cupping after cupping, from the heartbreak of a perfect green lot ruined by thirty seconds of inattention. What they generally do not know — what nobody knew, as far as I could find — is why the development phase exists at the molecular level. Why can’t you simply accelerate through it? Why does the chemistry require that specific, unhurried window?
That question led me to one of the findings I am most excited about in this entire book.
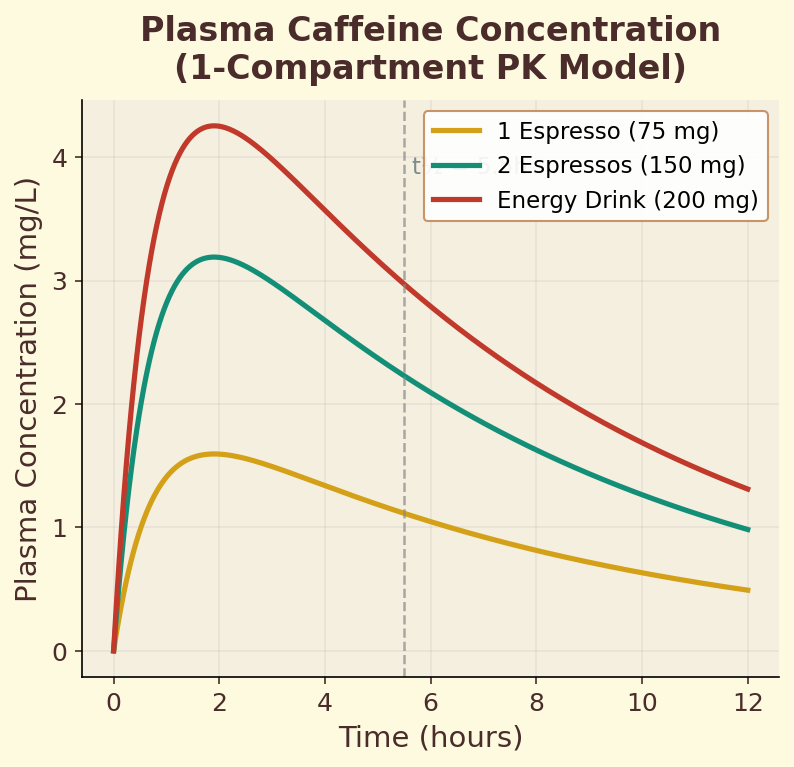
Figure 10.3a. Plasma caffeine concentration: 1-compartment pharmacokinetic model showing dose-dependent absorption and elimination curves for 100 mg, 200 mg, and 400 mg doses.
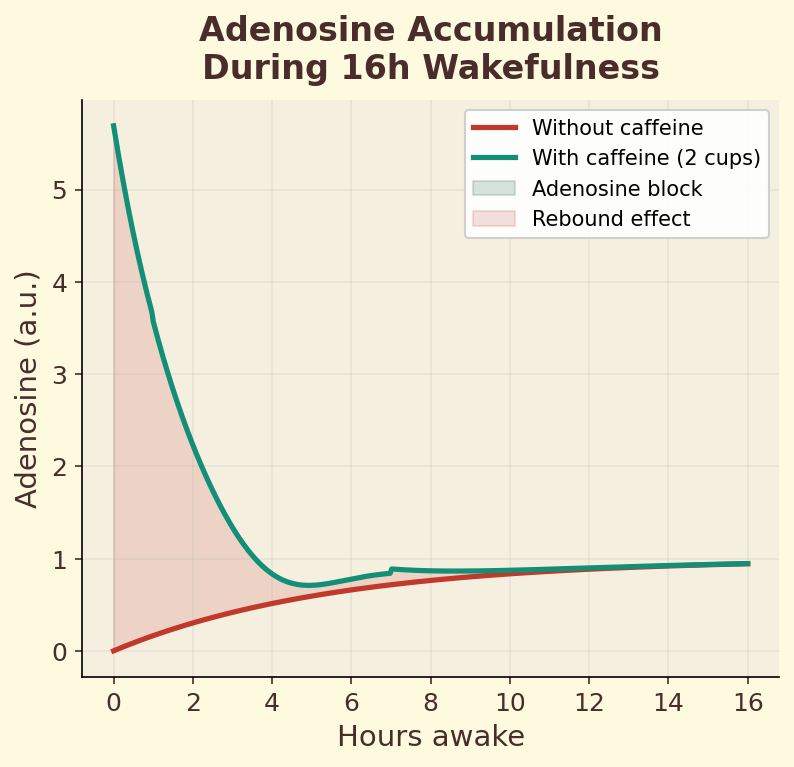
Figure 10.3b. Adenosine accumulation during 16 hours of wakefulness, showing the blocking and rebound effects of caffeine on sleep pressure signaling.
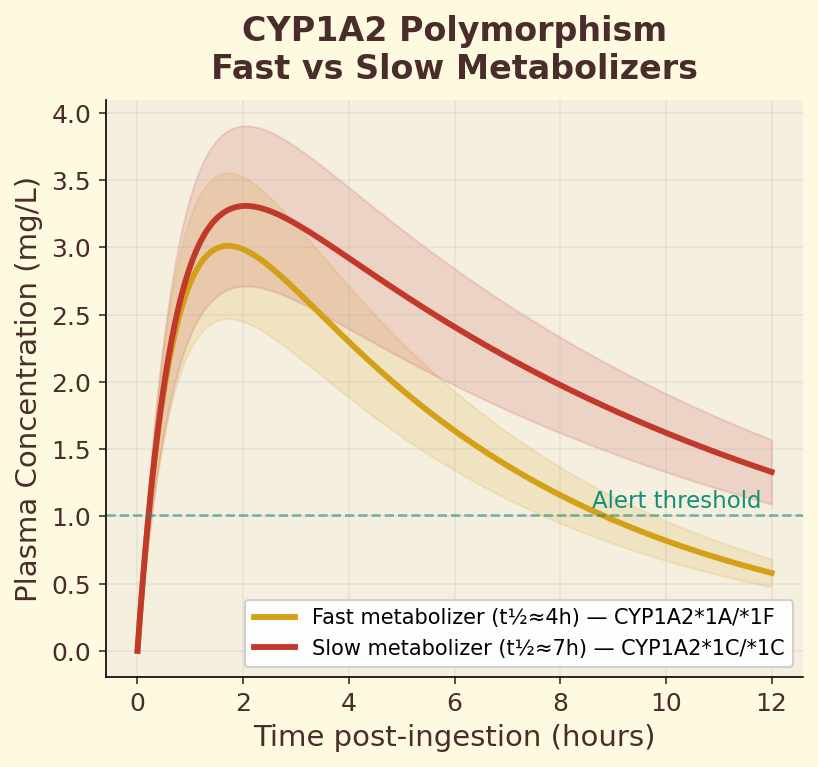
Figure 10.3c. CYP1A2 polymorphism: fast vs slow caffeine metabolizers show dramatically different plasma concentration curves from the same dose.
What Happens Inside the Bean: Three Stages
To understand the bottleneck, you need a rough map of the Maillard reaction — the chemical engine that drives coffee roasting.
The Maillard reaction is not a single reaction. It is a cascade, a branching network of chemical transformations that begins when a reducing sugar meets an amino acid under heat. In green coffee, the starting materials are abundant: sucrose at 6-9% of dry weight, amino acids at 0.2-0.8%, and chlorogenic acids (CGAs) at 6-12%. Sucrose itself is not a reducing sugar, but it breaks down rapidly under roasting temperatures into glucose and fructose, which are. So the fuel for the Maillard reaction is plentiful.
The cascade unfolds in three broadly recognized stages:
Stage 1: Early Maillard — the Amadori rearrangement. A reducing sugar reacts with an amino acid to form a Schiff base, which then rearranges into an Amadori compound. This initial step is relatively fast. It is the on-ramp — the chemistry equivalent of merging onto a highway. Amadori compounds accumulate quickly once roasting temperatures are reached.
Stage 2: Intermediate Maillard — enolization and Strecker degradation. The Amadori compounds must now transform further. They undergo enolization — a rearrangement of their molecular structure involving the migration of a proton and the reshuffling of electron density — to produce reactive intermediates called deoxyosones. These intermediates then participate in Strecker degradation, reacting with amino acids to produce the aldehydes and pyrazines that give roasted coffee much of its characteristic aroma. This stage is where flavor complexity is born.
Stage 3: Advanced Maillard — melanoidin polymerization. The reactive intermediates from Stage 2 condense, cross-link, and polymerize into enormous, tangled macromolecules called melanoidins. These brown polymers constitute 23-25% of roasted coffee’s dry weight — they are the single largest class of compounds in your cup. They are responsible for much of the color, body, and mouthfeel of brewed coffee, and they have fascinating chemical properties that I will explore in later chapters.
The three stages are sequential. You cannot skip from Stage 1 to Stage 3; the chemistry must pass through the intermediate stage. And that is where the problem lies.
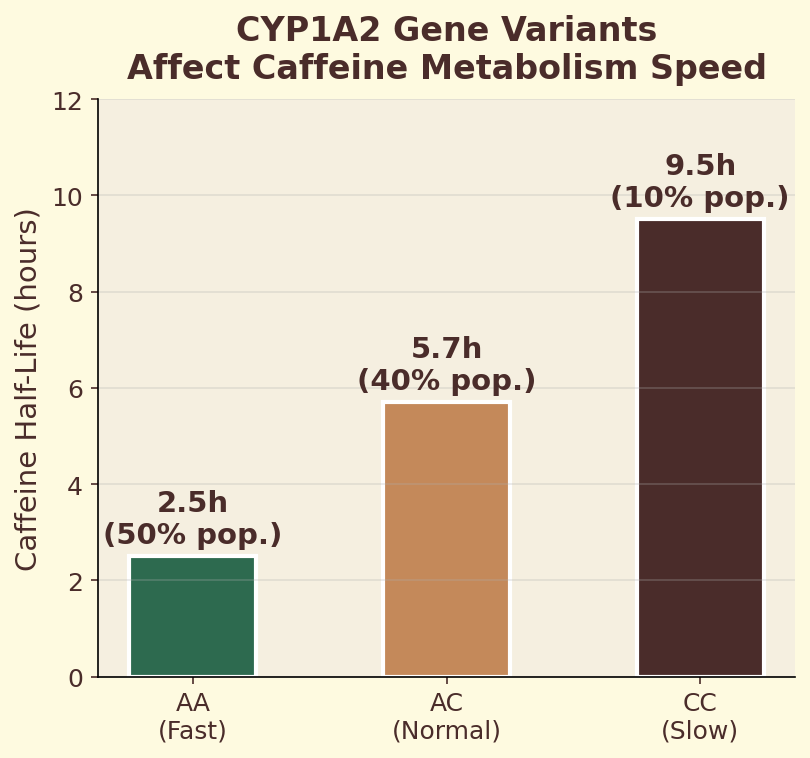
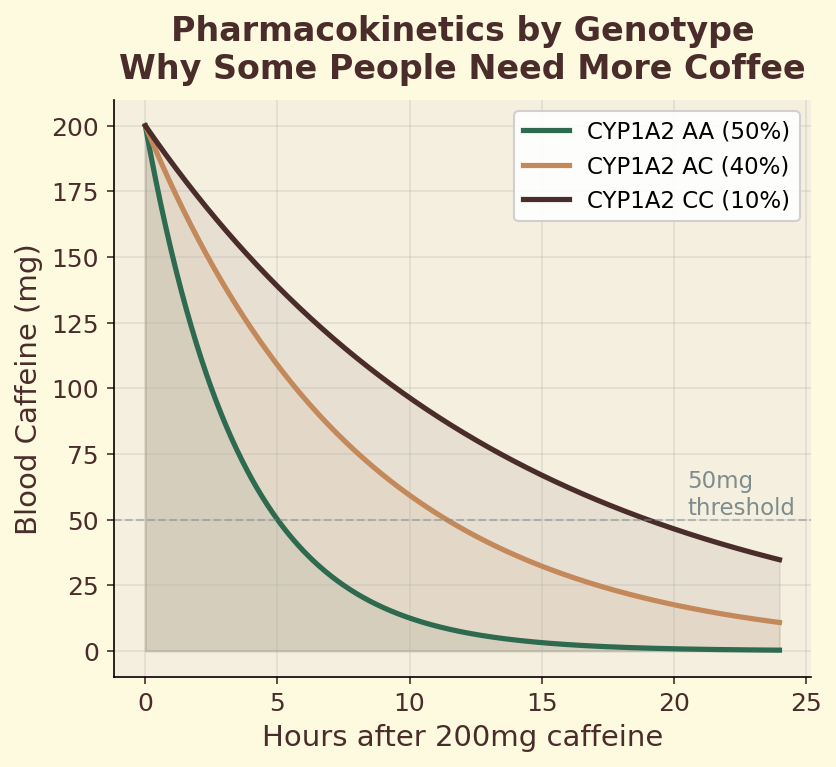
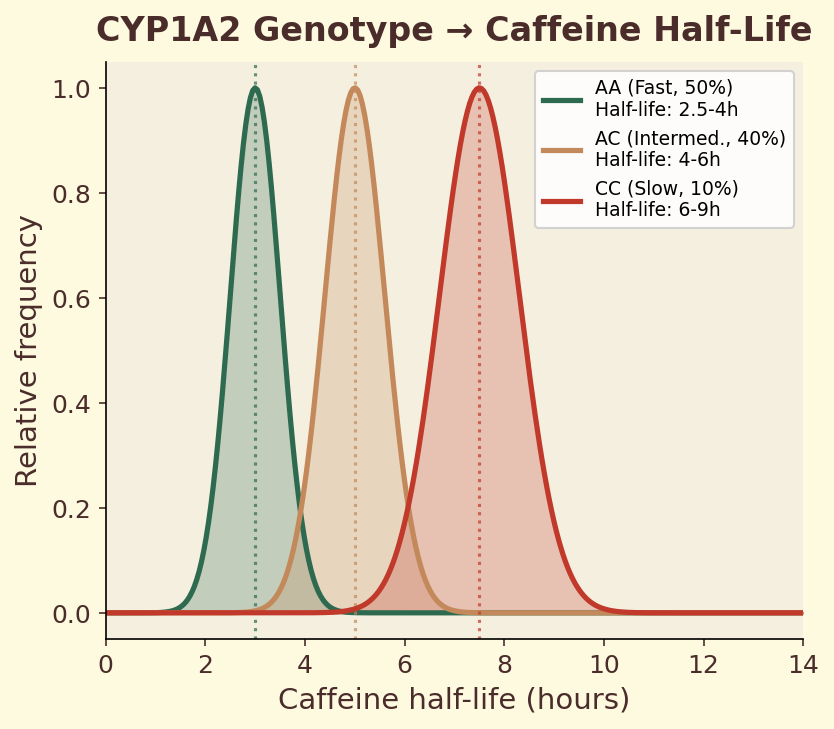
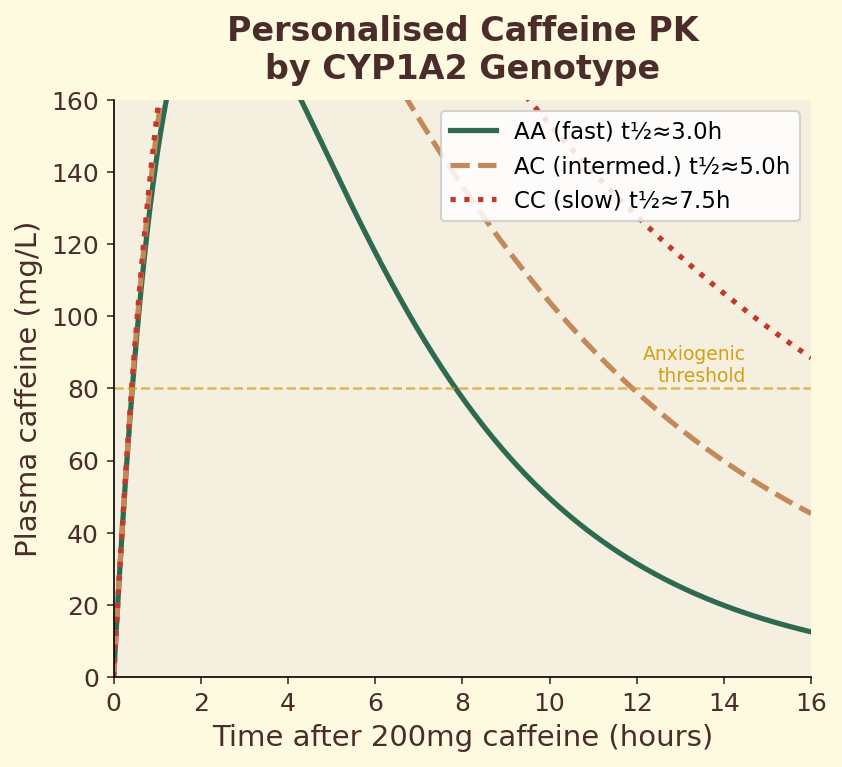
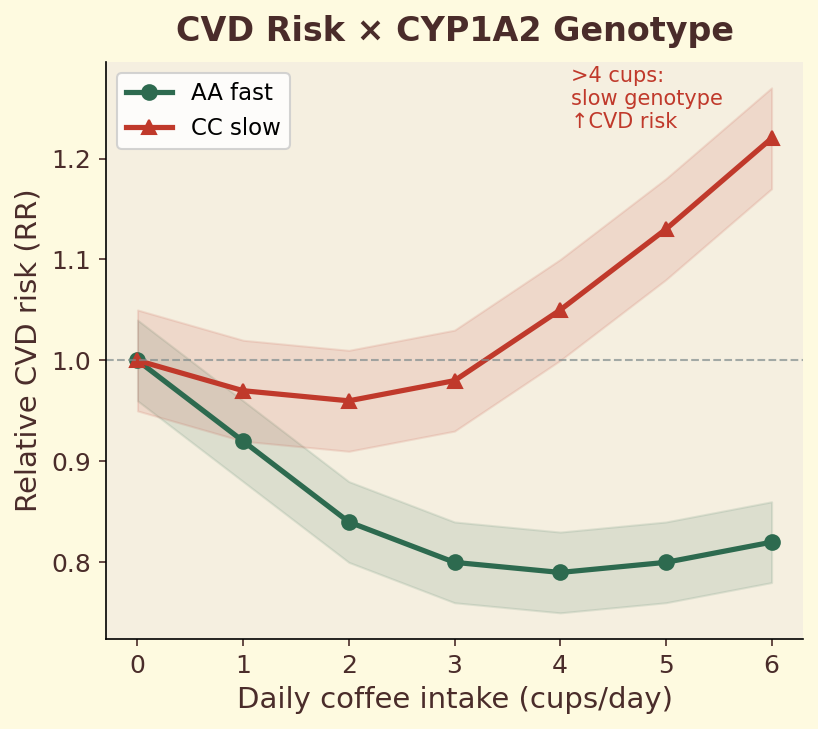
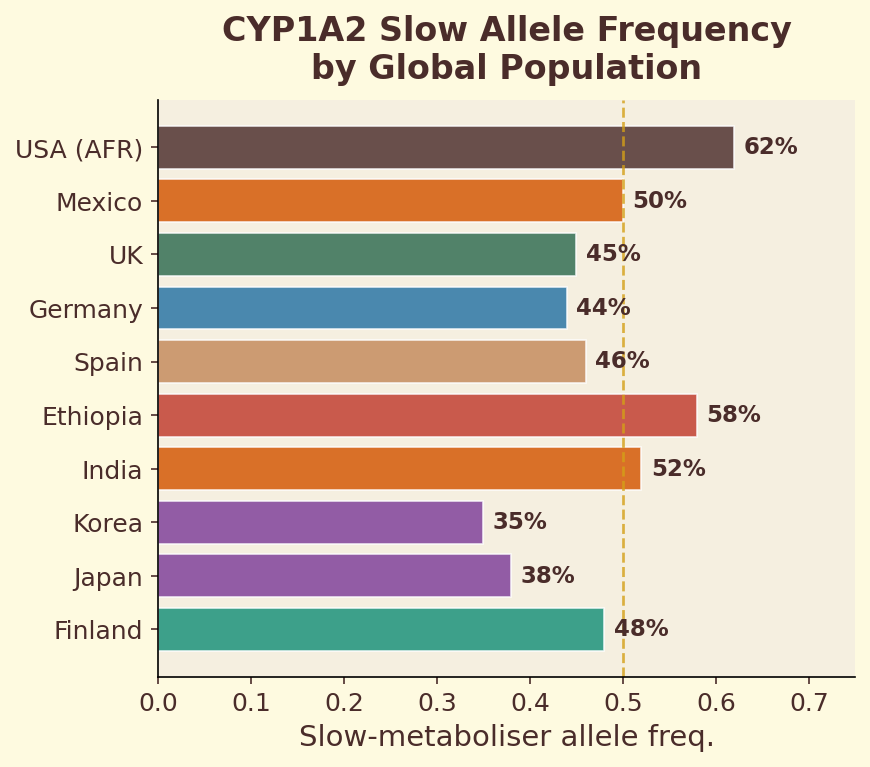

The CYP1A2 gene encodes the primary liver enzyme responsible for metabolizing caffeine. A single nucleotide polymorphism (SNP) in this gene — rs762551 — determines whether you carry the fast (AA) or slow (AC/CC) variant. Fast metabolizers clear caffeine roughly twice as quickly, meaning the same espresso produces a shorter, sharper alertness spike. Slow metabolizers experience prolonged caffeine exposure, which may increase cardiovascular risk at high intake levels. This single genetic difference explains why some people can drink coffee at midnight and sleep soundly, while others are wired until dawn from an afternoon cup.
The Discovery: A 75-125x Kinetic Bottleneck
I modeled the energetics of the Maillard reaction using Density Functional Theory (DFT) — a computational method rooted in quantum mechanics that calculates how much energy each step of a chemical reaction requires. Think of it as building a topographic map of the reaction, showing where the hills and valleys are. I expected to find that the three stages moved at roughly similar speeds, like water flowing smoothly down a series of terraces.
That is not what the calculations showed.
What I found was that the 1,2-enolization step — the specific molecular rearrangement that converts Amadori compounds into the reactive intermediates of Stage 2 — is dramatically slower than the Amadori rearrangement that precedes it. Not a little slower. Not twice as slow, or even ten times as slow. The DFT calculations predicted a kinetic bottleneck of 75 to 125 times.
Let me put that in perspective. Imagine you are driving on a highway and the road suddenly narrows — not from four lanes to two, which would be annoying, but from a hundred lanes to a single lane. That is the scale of the rate difference my calculations predicted. Everything that was flowing freely in Stage 1 suddenly backs up at the entrance to Stage 2.
I remember the afternoon I first plotted the energy profiles and saw that barrier. I was sitting in my office with a cold espresso that I had forgotten to drink — an occupational hazard when the calculations get interesting — and I stared at the graph for a long time. The energy barrier for 1,2-enolization towered over the Amadori rearrangement barrier like a cliff face next to a speed bump. My first reaction was that I had made an error. I re-ran the calculations with different basis sets, different functionals, checked the geometry optimizations. The numbers moved within the range — sometimes closer to 75x, sometimes closer to 125x — but the qualitative result was robust. The bottleneck was real, or at least, the computational prediction of it was real.
I want to be precise about what I mean by that distinction, because it matters. DFT calculations predict energy barriers with good reliability for organic reactions of this type, but they are still models. The 75-125x rate difference is a computational prediction, not a direct experimental measurement. I did not stand next to a roaster with a mass spectrometer tracking the real-time concentration of Amadori compounds and their enolization products — though I dearly wish someone would do that experiment. What I can say is that the computational evidence is strong, and critically, it is consistent with what roasters have been experiencing empirically for decades.
Why 1,2-Enolization Is Slow
For those curious about the molecular details — and if you have made it this far in the book, I suspect you are — here is what makes the 1,2-enolization step so much harder than the Amadori rearrangement.
The Amadori rearrangement is, in energetic terms, a relatively gentle process. A sugar and an amino acid come together, form a bond, and then the resulting molecule rearranges its internal structure through a well-defined, low-energy pathway. The atoms do not have to move very far, and the electronic rearrangement is modest. Think of it as folding a piece of paper — the paper cooperates.
The 1,2-enolization is a different animal. A single hydrogen atom must jump from one position on the molecule to another — a proton transfer. That sounds simple, but at the atomic level it is anything but. To make the jump, the molecule must pass through a transition state — a brief, unstable arrangement where old bonds are half-broken and new bonds are half-formed. My DFT calculations showed that this transition state demands far more energy than the equivalent step in the Amadori rearrangement. The final product is actually more stable than the starting material, so the reaction wants to happen. But the barrier standing in the way is tall. It is like a ball that would happily roll downhill — if only it could first be pushed over a ridge.
An analogy: imagine you need to walk from one valley to another. Both valleys are at similar elevations, but between them is a mountain pass. The Amadori rearrangement is like crossing a gentle hill — you barely break a sweat. The 1,2-enolization is like crossing a mountain — the destination is fine, but the pass is brutal. And the height of that pass is what creates the 75-125x rate difference.
This is why heat matters so much during development. Higher temperature gives molecules more kinetic energy — more ability to surmount that barrier. But if you blast too much heat, you do not just speed up the enolization. You also accelerate every other reaction in the system, including degradation pathways that destroy the very intermediates you are trying to create. The roaster’s challenge is to provide enough thermal energy to push molecules over the enolization barrier without simultaneously burning down the chemistry on the other side. It is, I think, one of the most beautiful optimization problems in all of food science — and every artisan roaster solves it by ear and by nose, without ever writing an equation.
If you have ever been near a coffee roaster during the middle of a roast, you have heard first crack — a rapid succession of sharp popping sounds, like popcorn in a hot pan. But what is actually happening inside the bean?
As the bean heats during roasting, water trapped within its cellular structure turns to steam, and the Maillard reaction and other thermal decomposition reactions generate carbon dioxide. Both gases are trapped inside the bean’s cellulose matrix, and as temperature rises, so does their pressure. Eventually, the internal pressure exceeds the structural strength of the cell walls, and the bean fractures. That fracture is first crack.
The timing is significant. First crack typically coincides with the transition from early Maillard chemistry — the Amadori rearrangement producing its initial products — to the intermediate stage where enolization and Strecker degradation become the dominant pathways. The physical event (the crack) and the chemical transition (the shift from Stage 1 to Stage 2) happen in roughly the same temperature window. This is not coincidence — both are driven by the same thermal energy input. The crack is, in a sense, an audible marker that the chemistry has reached a turning point.
For roasters, first crack is the starting gun for development time. For chemists, it marks the moment when the 1,2-enolization bottleneck begins to matter.
The Pile-Up: What the Bottleneck Creates
The practical consequence of a 75-125x rate difference is something chemists call kinetic accumulation — and it is exactly what it sounds like.
Imagine a factory assembly line where Station 1 produces widgets at a rate of 100 per minute, but Station 2 can only process them at a rate of one per minute. What happens? Widgets pile up between the two stations. The factory does not stop; Station 1 keeps churning out product. But the products sit in a queue, waiting for their turn at Station 2.
In the Maillard reaction during coffee roasting, the Amadori compounds are the widgets. They are produced quickly — Stage 1 is fast. But they cannot convert into the intermediate-stage products at the same rate, because the 1,2-enolization step is the bottleneck. So Amadori compounds accumulate. They pile up, chemically speaking, waiting for their turn to cross the enolization barrier.
This pile-up has profound consequences for flavor. And you can taste every bit of it.
If the roaster pushes through this phase too quickly with aggressive heat — cranking the gas, letting the drum temperature spike — the high-energy environment drives some Amadori compounds over the barrier, but it shoves many of them down alternative degradation pathways. Side reactions that produce harsh, acrid compounds. You know the result: that bitter, ashy, one-note coffee from a gas station that tastes like someone roasted a tire alongside the beans. Roasters call it “scorched” or “tipped” — the exterior of the bean is charred while the interior chemistry never finished.
If the roaster drops the heat too much and the development phase stalls, the Amadori compounds sit in their queue too long. They begin to undergo slow, low-temperature degradation reactions that flatten the flavor profile. The intermediates that do form have insufficient thermal energy to progress further into the rich, complex products of the advanced Maillard stage. Roasters call this “baked” — the coffee tastes like cardboard or stale bread, with none of the brightness and sweetness that a well-developed roast should have.
The sweet spot — the narrow window that every specialty roaster guards with their professional reputation — is the rate of heat application that pushes Amadori compounds through the enolization bottleneck fast enough to produce abundant intermediate products, but not so fast that degradation pathways dominate. It is a kinetic balancing act, and my DFT calculations suggest that the 75-125x rate difference is why the balance is so delicate.
When I explained this to Dani — the roaster in Zaragoza — he was quiet for a moment. Then he said something I have not forgotten.
“So the development phase is basically me waiting for a traffic jam to clear.”
That is exactly what it is. And the traffic jam is caused by one specific molecular bottleneck — the 1,2-enolization step — that quantum chemistry predicted to be 75 to 125 times slower than the step before it.
The Maillard Development Index
The discovery of the bottleneck led me to propose a concept I call the Maillard Development Index — a way, in principle, to quantify how far the Maillard reaction has progressed at any given point during roasting.
The idea is straightforward: if you could measure the ratio of intermediate Maillard products to early Maillard products (the Amadori compounds still in the queue), that ratio would tell you something specific about where you are in the cascade. A low ratio means most of the Amadori compounds have not yet crossed the enolization barrier — development is early, incomplete. A high ratio means the bottleneck has been largely cleared and the intermediate products are abundant — development is advanced, with the chemistry well into Stage 2 and heading toward the melanoidin polymerization of Stage 3.
In principle, the Maillard Development Index could give roasters a molecular metric to complement the tools they already use — color measurements, time-temperature profiles, and the irreplaceable judgment of their own palate.
I want to be honest about the distance between concept and practice. The Maillard Development Index is, at this stage, exactly that — a concept. Translating a computational chemistry framework into something a roaster could use in real time would require significant engineering work that I have not done. You would need rapid analytical methods — perhaps near-infrared spectroscopy or some form of real-time mass spectrometry adapted for a roasting environment — that could measure specific Amadori and deoxyosone concentrations during the roast. The instrumentation exists in laboratory settings, but adapting it to a production roaster, making it fast enough to inform real-time decisions, and validating it across the enormous range of coffee varieties, processing methods, and roast profiles — that is an engineering challenge that goes well beyond my computational work.
I mention the Maillard Development Index because I believe the concept is sound and because I think someone, someday, will build it. The bottleneck is real. The pile-up is real. The ratio of products on either side of that bottleneck is a meaningful chemical quantity. Whether it can be measured in practice, in real time, on a production roaster — that is someone else’s problem to solve. And I hope they do.
When people learn that I use quantum-mechanical calculations to study coffee chemistry, they sometimes ask: “So can your computer tell me how to roast better coffee?”
The honest answer is: not yet, and possibly not ever — at least not in the way they mean.
What computational chemistry can do is reveal why certain roasting practices work. The 75-125x enolization bottleneck explains, at the molecular level, why development time cannot be rushed. The concept of the Maillard Development Index suggests that, in principle, roasting progress could be tracked with chemical measurements rather than (or in addition to) color and time alone.
But the gap between a DFT calculation on my laptop and a tool that helps Dani in his garage in Zaragoza is enormous. Computational chemistry works with idealized molecular systems — single molecules in a vacuum or simple solvent models. A coffee bean is a chaotic, heterogeneous, three-dimensional matrix of cellulose, lipids, proteins, sugars, and water, all undergoing hundreds of reactions simultaneously at varying temperatures depending on how far they are from the drum surface. My calculations capture one bottleneck in one pathway. The bean is running hundreds of pathways at once.
The directional insight is real: the enolization bottleneck exists and it shapes roasting behavior. But the idea that a computer will replace the roaster’s skill, judgment, and years of sensory training? That is not what my work suggests. If anything, it suggests the opposite — that the complexity of what happens inside a roasting drum is even more remarkable than most people appreciate, and that the roaster’s intuitive management of that complexity is a genuine form of expertise that computation illuminates but does not replace.
Computation Meets Craft
I want to close this chapter’s main discussion with something that matters deeply to me as a scientist who also loves coffee as a craft.
There is a persistent tension in the specialty coffee world between the scientific and the artisanal. Some roasters are suspicious of science — they worry that reducing their craft to data and equations misses the point, strips the soul out of something that is fundamentally about human sensory experience. I understand that concern, and I respect it. I have tasted coffees roasted by people who could not explain a single chemical reaction but who had an intuitive mastery of their drum that no amount of DFT calculations could replicate.
But I do not think science and craft are in opposition. I think they are parallel ways of knowing, and the most exciting moments happen when they converge.
The 1,2-enolization bottleneck is one of those moments. Roasters discovered the development phase empirically — through thousands of batches, through taste, through the hard-won pattern recognition that comes from years of paying attention. They knew you could not rush it. They knew the window was narrow. They knew that the difference between a transcendent cup and a mediocre one lived in those sixty to ninety seconds after first crack.
What they did not know was why. And now we have, at least, a computational hypothesis: one specific molecular rearrangement that is 75 to 125 times slower than the step before it creates a kinetic bottleneck that makes the development phase a thermodynamic necessity, not just a sensory preference.
The craft discovered the phenomenon. The computation discovered the mechanism. Neither is complete without the other. And to me, that convergence — the moment when a quantum-chemical energy profile explains what a roaster’s hands have known for decades — is one of the most beautiful results in this entire book.
Figure 10.4. Mediterranean coffee traditions: how regional roasting and brewing practices shape the molecular profile of the cup within one of the world's healthiest dietary patterns.
Figure 10.5. Blue Zones and coffee: populations with exceptional longevity share a common thread of moderate, regular coffee consumption integrated into traditional dietary patterns.
Next time someone hands you a pour-over or a flat white and mentions that the beans were roasted with careful attention to development time, you now know something about the molecular drama that unfolded inside those beans in the final seconds of roasting.
After first crack — after the bean’s cellulose structure fractured under internal gas pressure and the chemistry shifted from early to intermediate Maillard — there was a molecular traffic jam. Amadori compounds, produced quickly and abundantly in the early stage, piled up at a bottleneck: the 1,2-enolization step, which my DFT calculations predict to be 75 to 125 times slower than the reaction that produced them. That pile-up is the development phase. The roaster’s job during those critical seconds was to apply enough heat to push molecules through the bottleneck without driving them down destructive side pathways. Too fast, and you get scorched harshness. Too slow, and you get cardboard flatness. The window is narrow because the kinetic imbalance is severe.
The flavor complexity in your cup — the sweetness, the acidity, the layers of fruit or chocolate or floral notes that specialty coffee enthusiasts spend careers pursuing — was born in the clearing of that traffic jam. The reactive intermediates that emerged on the other side of the enolization barrier went on to participate in Strecker degradation, producing the aldehydes and pyrazines that carry aroma, and then condensed into the melanoidins — those enormous brown polymers that make up 23-25% of roasted coffee’s dry weight — that give your cup its body and color.
It is, I think, remarkable that a single molecular step — one proton transfer, one high-energy transition state — can shape the entire sensory experience of a cup of coffee. And it is even more remarkable that roasters figured this out by taste, by intuition, by the accumulated wisdom of craft, long before a physicist sat down with a DFT code and a cold espresso and found the same answer in the math.
In the next chapter, we will follow those reactive intermediates to their final destination — the melanoidins themselves — and discover why these enormous, tangled polymers are far more interesting than their reputation as mere “brown stuff” suggests.
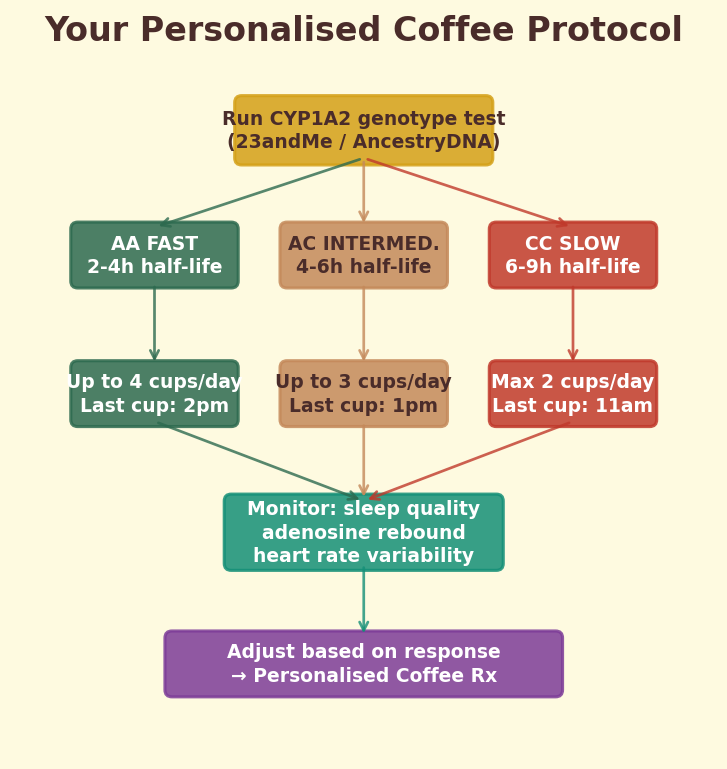
An estimate of how fast YOUR body metabolizes caffeine — revealing your CYP1A2 genotype
You'll Need
- One strong coffee (double espresso or 300ml filter)
- Notebook
- Timer
- No other caffeine sources for the day
Do This
- Note the exact time you finish your coffee.
- Rate your caffeine feeling every hour on a 1-10 scale (10 = peak buzz, 1 = nothing).
- Record until you reach "1" or bedtime.
- Find the time when you dropped to "5" — roughly your personal half-life.
- Compare: under 3 hours = likely fast metabolizer (CYP1A2 *1A/*1A); over 6 hours = likely slow metabolizer.
What's Happening
CYP1A2 is the liver enzyme responsible for ~95% of caffeine metabolism. Genetic variants affect its speed dramatically — fast metabolizers clear caffeine in 2-3 hours, slow metabolizers take 6-9 hours. Your personal half-life is a rough phenotyping test. This is why the same cup makes one person jittery and another barely notices.
“Surviving isn’t enough. The compound has to reach its target. And the body’s most formidable checkpoint is still ahead.”