Chapter 1: What's Really in Your Coffee?
Part I: The Molecules in Your Cup
Key Points
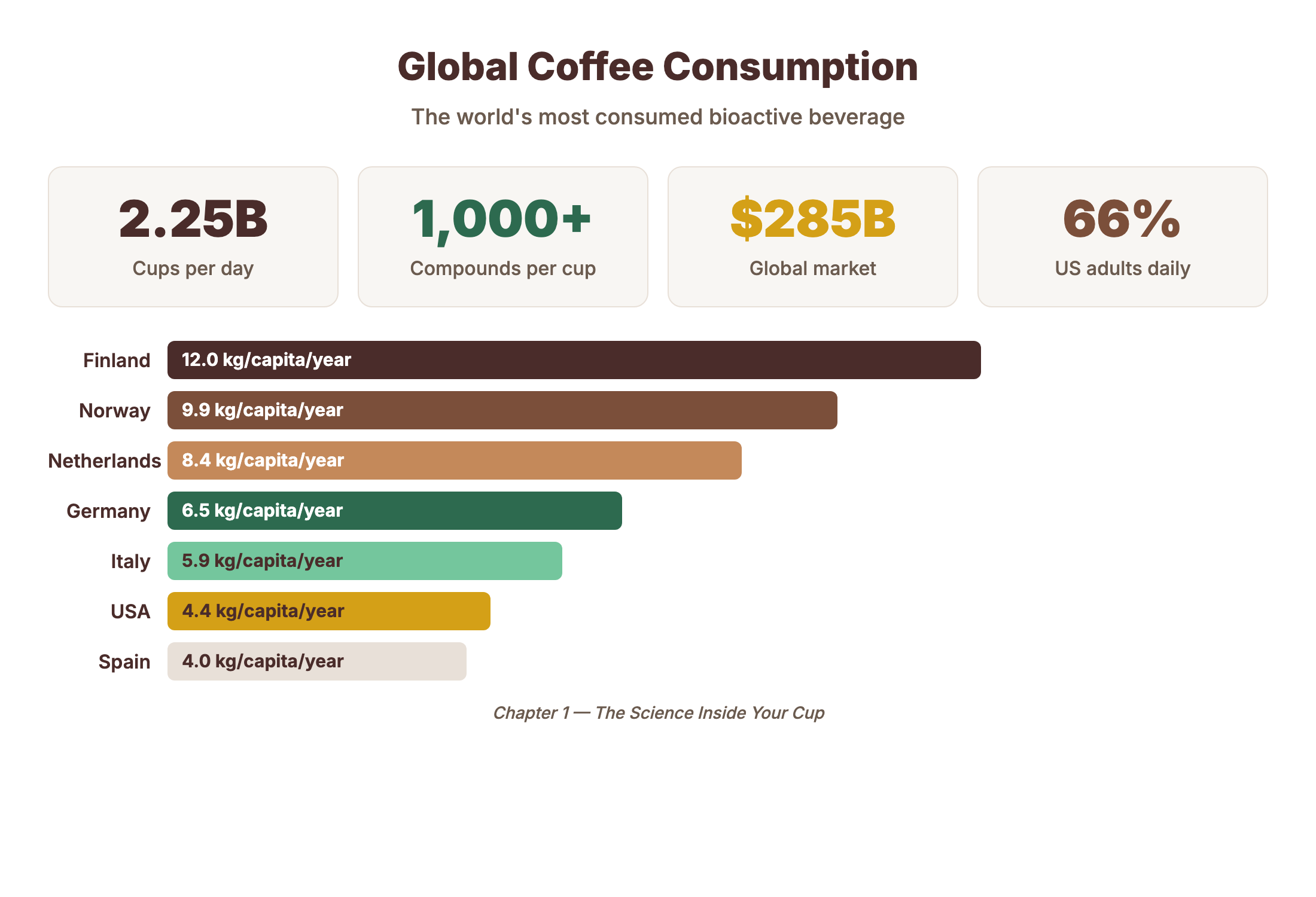
- Coffee contains 1,000+ identified chemical compounds — 15 key bioactives drive most effects
- Chlorogenic acids (CGAs) make up 6-12% of green bean dry weight and drive perceived acidity
- Melanoidins constitute ~25% of brewed coffee solids — the largest single compound class
- Trigonelline breaks down during roasting to produce niacin (vitamin B3): 10-40 mg per cup
- Your paper filter removes diterpenes — it is effectively a pharmacological intervention
I’m sitting at a specialty coffee shop in Barcelona’s Born district on a Tuesday morning, watching the barista work. She’s pulling an espresso — an Ethiopian single-origin, natural process, light roast — and she’s doing it with the kind of focused precision I recognize from my own years at the bench. The grind is dialed in. The water temperature is exact. She watches the stream like a scientist watching a chromatography column, waiting for the color to shift.
I smile. She’s not wrong. Those flavor descriptors correspond to real, identifiable volatile compounds — ethyl 3-methylbutanoate for the blueberry, a cluster of pyrazines for the chocolate, linalool for the jasmine. I know this because I’ve spent a significant part of my career studying how molecules behave in biological systems, and coffee — this deceptively simple beverage — is one of the most staggeringly complex chemical mixtures that humans voluntarily put into their bodies every single day.
That little cup she just poured? It contains over 1,000 identified chemical compounds. More than wine. More than chocolate. More than most pharmaceutical formulations. And every one of those compounds is doing something once it enters your body — binding to a receptor, modulating an enzyme, interacting with your gut microbiome, crossing or failing to cross the blood-brain barrier.
The barista sees flavor. I see a pharmacological event.
This book is about what I see.
Coffee’s Molecular Census
Let me start with a number that still impresses me, even after years of working in computational chemistry: researchers have identified over 1,000 distinct chemical compounds in brewed coffee. Some estimates push this higher — into the range of 1,500 or more — depending on how you define the boundaries of detection and whether you include the full spectrum of volatile aromatics captured by modern headspace analysis.
To put this in perspective, wine — which has its own rich tradition of chemical analysis and sensory science — contains roughly 600 to 800 identified compounds. Dark chocolate comes in around 600. Tea, depending on the variety and preparation, yields between 400 and 600. Coffee surpasses them all.
This makes coffee one of the most chemically complex beverages that humans consume. And I want to be precise about why that matters, because it’s not just a fun fact for cocktail parties.
When you have a system with over 1,000 active components, you cannot understand it by isolating one molecule at a time. This is the fundamental limitation of the reductionist approach that dominated nutritional science for decades — the idea that you could extract caffeine from coffee, study caffeine in a cell culture, and then claim you understood what coffee does to the human body. You can’t. Because caffeine in isolation behaves differently from caffeine in the presence of chlorogenic acids, which behave differently in the presence of melanoidins, which behave differently depending on whether cafestol and kahweol made it through your paper filter.
Coffee is not a drug. It is a multi-component pharmacological system. And understanding it requires tools that can handle that complexity. Tools that, for most of human history, we simply didn’t have.
We have them now.
The Big 15: Bioactive Compounds That Matter Most
Of those 1,000-plus compounds, not all carry equal weight when it comes to your health and your sensory experience. Research over the past three decades has identified approximately 15 bioactive compounds — or compound families — that account for the majority of coffee’s measurable biological effects and its characteristic flavor profile. I think of them as the Big 15. Let me walk you through them.
1. Caffeine (1,3,7-trimethylxanthine)
The famous one. The reason most people drink coffee in the first place. Caffeine is a purine alkaloid with the molecular formula C₈H₁₀N₄O₂ and a molecular weight of 194.2 g/mol. It constitutes roughly 1-2% of the dry weight of Arabica beans and up to 2.7% in Robusta.
Its primary mechanism of action is well established: caffeine is an adenosine receptor antagonist. Adenosine is a neurotransmitter that accumulates in your brain over the course of waking hours, progressively signaling drowsiness by binding to A₁ and A₂A receptors. Caffeine’s molecular structure is similar enough to adenosine that it occupies these same receptors — but without activating them. It blocks the drowsiness signal without replacing it with anything. The result is that other stimulatory neurotransmitters — dopamine, norepinephrine, glutamate — continue operating without adenosine’s inhibitory brake.
This is elegant. It’s also just the beginning of caffeine’s story, because adenosine receptors are found throughout the body, not just the brain. Research suggests that caffeine’s effects on cardiovascular function, exercise performance, and even certain aspects of metabolic regulation may all trace back to this single mechanism operating across different tissue types.
But here’s the thing about caffeine that most people miss: it’s not the most abundant bioactive compound in your cup. It’s not even close.
2. Chlorogenic Acids (CGAs)
If caffeine is coffee’s celebrity, chlorogenic acids are its workhorse. CGAs constitute 6-12% of the dry weight of green (unroasted) coffee beans, making them among the most abundant polyphenols in the human diet for regular coffee drinkers. A single cup of coffee can deliver 70-350 mg of chlorogenic acids, depending on the bean variety, roast level, and brewing method.
The term “chlorogenic acid” is actually a family name. It encompasses dozens of isomers — esters formed between hydroxycinnamic acids (like caffeic acid and ferulic acid) and quinic acid. The major players include 5-caffeoylquinic acid (5-CQA), which is often what people mean when they say “chlorogenic acid” without specifying, along with 3-CQA, 4-CQA, and the dicaffeoylquinic acids (3,5-diCQA, 4,5-diCQA, and 3,4-diCQA). Each isomer has a slightly different biological profile.
CGAs are potent antioxidants — they scavenge free radicals and chelate pro-oxidant metal ions. But research suggests their biological relevance extends well beyond simple antioxidant activity. Studies in cell models and animal models have associated CGAs with effects on glucose metabolism, blood pressure regulation, and neuroprotection, though I want to emphasize that translating these findings to firm conclusions about human health requires caution and more clinical data.
CGAs are also central to coffee’s flavor. They contribute to perceived acidity and, upon thermal degradation during roasting, generate some of the compounds responsible for bitterness and astringency. Their fate during roasting is one of the most consequential chemical stories in all of food science, and we’ll return to it shortly.
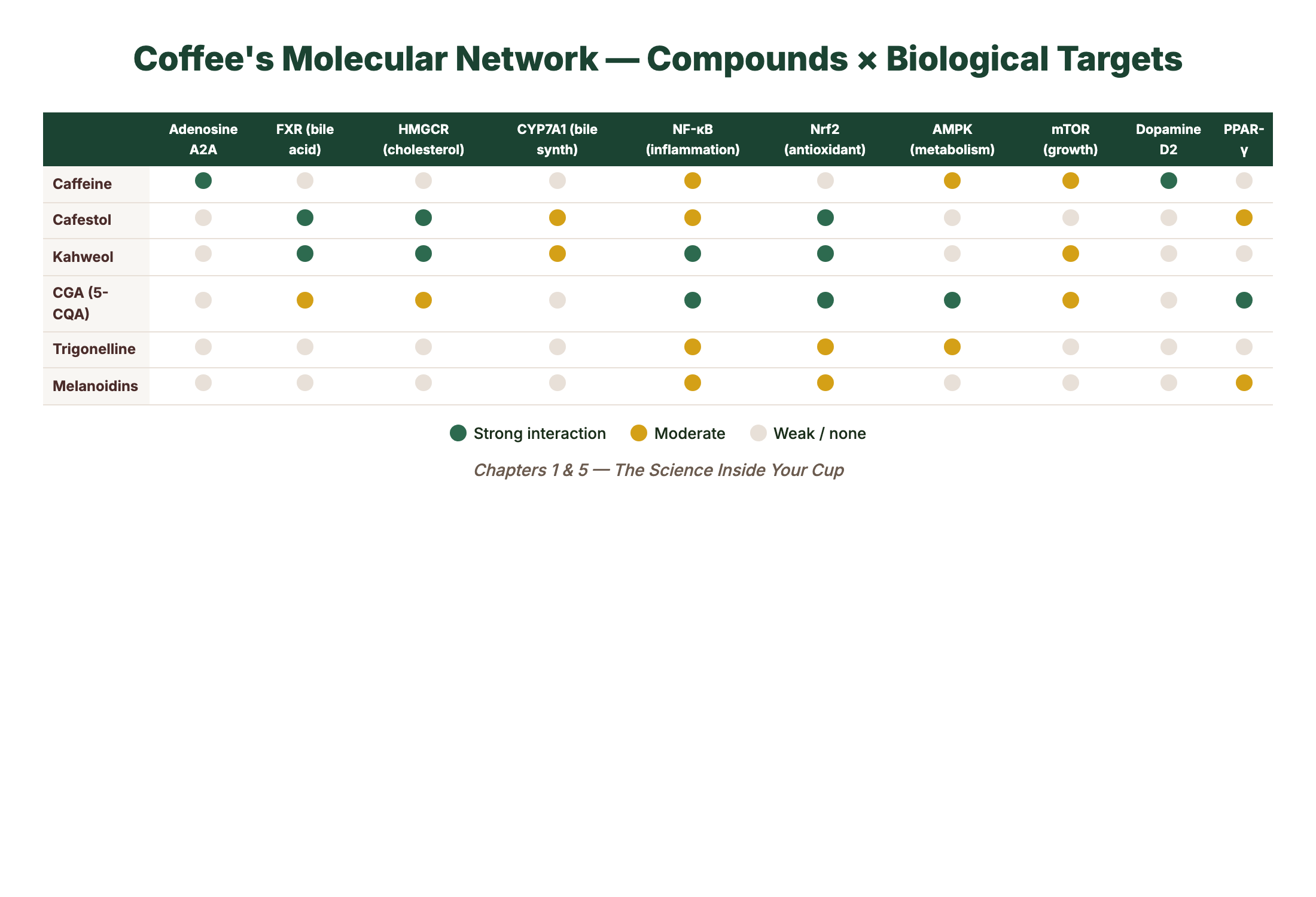
3. Cafestol (C₂₀H₂₈O₃, MW 316.4)
Cafestol is a diterpene — a class of compounds built from four isoprene units — found in the oily fraction of coffee beans. It is present at approximately 0.4-0.7% of dry weight in Arabica coffee. Along with its close relative kahweol, cafestol is concentrated in the lipid fraction of coffee and is primarily extracted into brewed coffee when no paper filter is used. This is why unfiltered coffee preparations — Turkish coffee, French press, Scandinavian boiled coffee, and espresso to a lesser degree — contain significantly more cafestol than drip-filtered coffee.
Cafestol is notable for having one of the most potent cholesterol-raising effects of any dietary compound identified in food. Research suggests that cafestol raises serum LDL cholesterol by suppressing bile acid synthesis via downregulation of cholesterol 7α-hydroxylase (CYP7A1) in the liver. Studies have estimated that consuming five cups of unfiltered coffee per day may raise LDL cholesterol by approximately 6-8 mg/dL.
The paper filter, it turns out, is not just a convenience. It is a pharmacological intervention.
4. Kahweol (C₂₀H₂₆O₃, MW 314.4)
Kahweol is cafestol’s molecular cousin. The two compounds are structurally almost identical — kahweol differs by having one additional double bond in its furan ring. This seemingly minor structural difference gives kahweol slightly different biological properties. Like cafestol, kahweol raises LDL cholesterol, though some research suggests it may also have anti-inflammatory and potentially hepatoprotective properties that partially offset this effect.
Kahweol is found almost exclusively in Arabica coffee. Robusta beans contain cafestol but very little kahweol, which is one of several chemical distinctions between the two major commercial species.
5. Trigonelline
Trigonelline is the second most abundant alkaloid in coffee after caffeine, present at roughly 0.6-1.0% of the dry weight of green beans. On its own, trigonelline contributes a slightly bitter taste. But its real significance emerges during roasting.
When subjected to the temperatures of the roasting process (typically 180-230°C), trigonelline undergoes thermal decomposition. One of its major breakdown products is nicotinic acid — better known as niacin, or vitamin B3. A single cup of coffee can provide 10-40 mg of niacin, which is a nutritionally meaningful amount (the recommended daily intake for adults is 14-16 mg). This makes coffee one of the most significant dietary sources of niacin in many populations, a fact that often surprises people.
Trigonelline’s decomposition also generates pyridines and pyrroles, which contribute to coffee’s characteristic roasted aroma. So when you smell that distinctly “coffee” smell as the beans come out of the roaster, part of what you’re detecting is the molecular remains of trigonelline.
6. Melanoidins
Here is perhaps the most underappreciated component of your daily cup. Melanoidins are high-molecular-weight brown polymers formed during the Maillard reaction — the complex cascade of chemical reactions between amino acids and reducing sugars that occurs during roasting. They are not present in green coffee at all. They are created entirely by the roasting process.
And they are not minor players. Melanoidins constitute approximately 23-25% of the dry weight of brewed coffee. Let me say that again: roughly a quarter of the dissolved solids in your coffee cup are melanoidins. This makes them, by mass, the single largest class of compounds in brewed coffee.
Despite their abundance, melanoidins remain poorly characterized compared to smaller, more tractable molecules like caffeine or CGAs. This is partly because they are structurally heterogeneous — no two melanoidin molecules are exactly alike, which makes them nightmarish to isolate and study using traditional analytical chemistry. They are defined more by their process of formation (Maillard reaction) and their physical properties (brown color, high molecular weight, solubility in water) than by a precise chemical structure.
Research suggests that melanoidins may have antioxidant activity, prebiotic effects in the gut, and metal-chelating properties. They also contribute significantly to the body and mouthfeel of coffee — that sense of weight and texture that distinguishes a full-bodied espresso from a thin, watery brew.
7. Caffeic Acid
Despite the name, caffeic acid is not derived from caffeine. It’s a hydroxycinnamic acid and one of the building blocks of chlorogenic acids. When CGAs break down during roasting or digestion, caffeic acid is one of the fragments released. It has demonstrated antioxidant properties in laboratory studies and is one of the most widely distributed phenolic compounds in the plant kingdom.
8. Ferulic Acid
Another hydroxycinnamic acid, closely related to caffeic acid but with an additional methyl group. Ferulic acid is released from chlorogenic acids during both roasting and gastrointestinal digestion. Research suggests it may have anti-inflammatory and UV-protective properties, though human clinical evidence remains limited.
9. Quinic Acid
The other half of the chlorogenic acid molecule. When CGAs break down, quinic acid is released alongside the hydroxycinnamic acid component. Quinic acid contributes to the perceived acidity of coffee and, at higher concentrations (typically in darker roasts where more CGAs have decomposed), can contribute to bitterness and astringency. It’s one of the reasons very dark roasts taste harsh to many people — the CGAs that provided a pleasant, bright acidity in lighter roasts have been converted into quinic acid and other degradation products.
10. N-methylpyridinium (NMP)
A compound formed during roasting from the decomposition of trigonelline. NMP is essentially absent in green coffee and increases with roast intensity. Research suggests it may stimulate gastric acid secretion less than other coffee components, and some studies have explored whether NMP-enriched coffees might be gentler on the stomach. The evidence is preliminary but intriguing.
11-15. The Supporting Cast
The remaining members of the Big 15 include 5-hydroxymethylfurfural (HMF), a Maillard reaction intermediate that has attracted attention for both its potential toxicity at high doses and its antioxidant activity at low doses; catechol, a simple diphenol with pro-oxidant properties; 4-vinylguaiacol, one of the key volatile phenols responsible for coffee’s spicy, clove-like aroma notes; theophylline, a dimethylxanthine structurally related to caffeine that is present in trace amounts and acts as a bronchodilator; and diterpene esters, various esterified forms of cafestol and kahweol that may have distinct biological activities from their free forms.
When you open a bag of freshly roasted coffee and that intoxicating aroma hits you, you’re experiencing one of the most complex scent profiles in the natural world. Researchers have identified over 800 volatile aromatic compounds in roasted coffee — more than in any other food or beverage.
These volatiles span nearly every major chemical class: pyrazines (nutty, earthy), furans (caramel, sweet), pyrroles (earthy, musty), thiophenes (meaty, savory), aldehydes (fruity, green), and ketones (buttery, caramel). Many are present at concentrations near or below the threshold of individual detection, but they interact synergistically, each one modulating the perception of others.
The remarkable thing is that none of these volatiles exist in the green bean. Every single one is created during roasting, through the Maillard reaction, Strecker degradation, caramelization, and thermal decomposition of precursors like trigonelline and chlorogenic acids. A green coffee bean smells grassy and slightly vegetal. The roasting process is, in a very real sense, the creation of “coffee” as a sensory experience.
Here’s what I find most fascinating from a computational perspective: we can now model the Maillard reaction pathways that generate these volatiles. We can predict which precursor compounds in a specific bean variety will yield which flavor molecules at a given roast temperature and duration. This is the kind of work that used to require thousands of hours of analytical chemistry. Today, our models can narrow the search space dramatically, guiding experimentalists toward the most promising conditions to test. The nose and the algorithm are learning to work together.
Why 1,000 Matters: Coffee as a Multi-Component System
I want to return to that number — over 1,000 compounds — and explain why it changes everything about how we should think about coffee and health.
In pharmaceutical science, we typically study drugs as single molecules. One compound, one target, one mechanism of action. Aspirin inhibits cyclooxygenase. Statins inhibit HMG-CoA reductase. This reductionist framework has been extraordinarily successful for developing medications, and it is the foundation of modern pharmacology.
But coffee is not a drug. It is a chemical ecosystem. And when you try to apply the single-compound paradigm to a 1,000-compound system, you run into problems very quickly.
Consider this: caffeine at the doses present in a typical cup of coffee has mild vasoconstrictive effects — it narrows blood vessels. Chlorogenic acids, at typical dietary doses, appear to have vasodilatory effects — they widen blood vessels. Both compounds are present simultaneously in every cup of coffee. What happens to your blood pressure? The answer is not “caffeine wins” or “CGAs win.” The answer depends on the ratio of the two, the presence of other modulatory compounds, your individual genetics (particularly your CYP1A2 genotype, which determines how fast you metabolize caffeine), your habitual coffee consumption, and probably a dozen other factors we haven’t fully mapped yet.
This is why the epidemiological literature on coffee and health has been so confusing for so long. For decades, studies seemed to contradict each other — coffee was protective, then it was harmful, then it was neutral, then it was protective again. The confusion arose, in part, because researchers were trying to understand a multi-component system using single-variable thinking.
The field that offers a way out of this impasse is called network pharmacology — the study of how multiple compounds interact simultaneously with multiple biological targets across multiple cellular pathways. Instead of asking “What does caffeine do?”, network pharmacology asks “What does the entire chemical profile of coffee do, acting on the entire network of relevant biological targets?”
This requires computational tools. There is simply no way to experimentally test every possible combination of 1,000 compounds against every possible biological target. The combinatorial space is too vast. But computational models — molecular docking, molecular dynamics simulations, machine learning classifiers, ADMET prediction algorithms — can explore this space in silico, generating hypotheses that experimentalists can then test in the lab.
That is the central promise of this book. Not that computers have replaced the laboratory, but that they have made it possible to ask questions that were previously unanswerable. We’ll explore network pharmacology in depth in Part II.
Green vs. Roasted: The Great Transformation
If you’ve ever held a green coffee bean in your hand, you know it looks nothing like the glossy brown object you drop into your grinder. It’s dense, hard, pale green or yellowish, and it smells like grass and hay. It tastes, if you’re brave enough to chew one, astringent and vegetal — nothing like coffee.
The transformation from green bean to roasted coffee is one of the most dramatic chemical metamorphoses in all of food processing. During a typical roast — which lasts anywhere from 8 to 20 minutes at temperatures between 180 and 230°C — the chemical profile of the bean is fundamentally restructured. Some compounds are destroyed. Others are created from scratch. Some are converted into entirely different molecules. Let me trace the major chemical storylines.
Sucrose Caramelizes
Green coffee beans contain 6-9% sucrose by dry weight — a surprisingly large amount. During roasting, this sucrose undergoes caramelization, a thermal decomposition process that breaks the disaccharide into glucose and fructose, which then undergo further reactions to produce caramel-flavored furanones, maltol, and a host of brown pigments. By the end of a medium roast, essentially all of the original sucrose has been consumed. It’s one of the major fuel sources for the entire cascade of roasting chemistry.
Chlorogenic Acids Degrade
Remember those CGAs that constitute 6-12% of the green bean? Roasting destroys them — progressively and dramatically. A light roast may retain 50-80% of the original CGA content. A medium roast retains roughly 30-50%. A dark roast? As little as 5-10% of the original CGAs may survive.
This has direct health implications. If you’re drinking coffee in part for its polyphenol content, roast level matters. Research suggests that light and medium roasts deliver significantly more chlorogenic acids per cup than dark roasts. The trade-off is that dark roasts generate more melanoidins and more NMP, which have their own distinct biological profiles.
The degradation of CGAs also transforms coffee’s flavor. Intact CGAs contribute a bright, pleasant acidity — the quality that specialty coffee professionals describe as “fruity” or “wine-like.” As they break down, they release quinic acid and caffeic acid, which at higher concentrations contribute bitterness and astringency. This is part of why dark roasts taste more bitter and less acidic than light roasts.
Melanoidins Form
As CGAs degrade and sucrose caramelizes, the Maillard reaction is simultaneously building something new. Amino acids in the bean react with reducing sugars to generate melanoidins — those high-molecular-weight brown polymers I described earlier. This is where the brown color comes from. This is where the body and mouthfeel come from. And this is where approximately 23-25% of the final dry weight of brewed coffee originates.
The Maillard reaction is not a single reaction. It is a cascade of hundreds of interconnected reactions, producing thousands of intermediate and final products. It is, in a sense, a chemical explosion in slow motion, and it is happening inside every coffee roaster in the world, right now, as you read this.
Trigonelline Converts to Niacin
As I mentioned earlier, roasting breaks down trigonelline into nicotinic acid (niacin) and a constellation of aromatic pyridines and pyrroles. This is a rare example of a food processing step that creates a vitamin. Most processing destroys nutrients. Coffee roasting actually generates one.
Hundreds of New Volatiles Appear
The most dramatic result of roasting, from a sensory perspective, is the creation of the volatile aromatic compounds that define coffee’s smell and taste. Over 800 volatiles have been identified in roasted coffee, and virtually none of them were present in the green bean. They are products of the Maillard reaction, Strecker degradation, caramelization, and the thermal decomposition of various precursor molecules.
The specific profile of volatiles — which ones are present and in what ratios — is determined by roast temperature, roast duration, rate of heat transfer, and the chemical composition of the starting bean. This is why an Ethiopian natural and a Colombian washed coffee taste so different even when roasted identically, and why the same bean can taste wildly different depending on roast profile. The starting chemistry matters. The process chemistry matters. Everything matters.
We’ll explore the computational modeling of roasting chemistry — including how machine learning is being applied to predict flavor outcomes from bean composition and roast parameters — in Part III.
Here’s something that puzzled researchers for years: decaffeinated coffee, in large epidemiological studies, appears to retain many of the health associations attributed to regular coffee. Research suggests that decaf drinkers show similar correlations with reduced risk of type 2 diabetes and liver disease as regular coffee drinkers, though the effect sizes are sometimes smaller.
If caffeine were the primary driver of coffee’s biological effects, this wouldn’t make sense. But it makes perfect sense when you remember the Big 15. Decaffeination removes roughly 97% of the caffeine, but it leaves most of the other bioactive compounds substantially intact. Chlorogenic acids, melanoidins, trigonelline, cafestol, kahweol — they’re all still there (some are modestly reduced depending on the decaffeination method, but the overall profile is preserved).
This is what I call the Caffeine Paradox: the most famous molecule in coffee may not be the most important one for health. Caffeine gets the headlines. The other 999 compounds may be doing the heavy lifting.
This realization has shifted the field’s attention. Increasingly, research is focusing on CGAs, melanoidins, and the coffee matrix as a whole, rather than studying caffeine in isolation. It’s a move away from reductionism and toward systems thinking. And it’s a move that computational tools — which can model multi-component interactions — are uniquely positioned to support.
Why Computational Tools Matter
I’ve spent the first part of this chapter describing what’s in your coffee — the molecular census, the Big 15, the transformation that roasting performs. All of this knowledge comes from decades of painstaking analytical chemistry: high-performance liquid chromatography (HPLC), gas chromatography-mass spectrometry (GC-MS), nuclear magnetic resonance (NMR) spectroscopy, and a battery of other techniques that can identify and quantify individual compounds.
This work is essential. But it answers only one question: what is present?
The question that matters more — the question that connects your morning cup to your afternoon energy, your cardiovascular health, your gut microbiome, your risk of neurodegenerative disease — is a different one: what does it do when it enters your body?
To answer that question, you need to know which of those 1,000 compounds are absorbed through the gut wall and which pass through undigested. You need to know which ones are metabolized by the liver’s cytochrome P450 enzymes and what metabolites they produce. You need to know which receptors they bind — not just their primary targets, but their off-targets, the unintended interactions that often drive both side effects and unexpected benefits. You need to know whether they can cross the blood-brain barrier. You need to know their half-lives, their tissue distribution, their protein binding affinities.
For a single drug molecule, answering these questions takes years of preclinical research and clinical trials. For 1,000 compounds simultaneously? That’s not possible through experiment alone.
This is where computational tools become not just helpful but essential.
Molecular docking can predict how a coffee compound fits into the binding pocket of a biological receptor — whether caffeine’s interaction with the adenosine A₂A receptor is stronger or weaker than a particular CGA isomer’s interaction with the same site.
Molecular dynamics simulations can model how these binding events play out over time, revealing whether a compound stays locked in place or dissociates quickly, and how the receptor’s shape changes in response.
ADMET prediction (Absorption, Distribution, Metabolism, Excretion, and Toxicity) uses machine learning models trained on thousands of known drugs to predict the pharmacokinetic fate of coffee compounds — which ones your body will absorb, how quickly it will eliminate them, and whether any metabolites might be problematic.
Network pharmacology integrates all of this information into a systems-level map, revealing how the multi-component mixture of coffee interacts with the multi-target network of human biology.

These tools don’t replace experiments. They guide them. They tell the experimentalist: “Of these 1,000 compounds, these 23 are the most likely to cross the blood-brain barrier. Start there.” They tell the epidemiologist: “If these three pathways are modulated simultaneously, you would expect to see this pattern in population data. Check whether you do.”
That’s what this book is about. Not coffee as a beverage, but coffee as a case study in how modern computational science can decode the complexity of what we eat and drink. Coffee is the ideal subject because it is complex enough to be interesting, well-studied enough to have benchmarks, and consumed by enough people that the stakes are genuinely high.
Let me take you back to that coffee shop in Barcelona.
The barista has pulled my shot. It’s the same Ethiopian natural she served the customer before me — notes of blueberry and dark chocolate, maybe a little jasmine if I let it cool. I take a sip.
Here’s what I know is happening.
Approximately 80-100 mg of caffeine are beginning to dissolve across the mucous membrane of my mouth and stomach lining, heading for my adenosine receptors. Within 15 minutes, my alertness will tick upward. Within 45 minutes, caffeine will reach peak plasma concentration.
Simultaneously, somewhere between 100 and 250 mg of chlorogenic acids are entering my digestive system. Some will be absorbed in the small intestine. Others will reach the colon intact, where my gut bacteria will cleave them into caffeic acid and quinic acid — metabolites with their own distinct biological activities.
The melanoidins — representing roughly a quarter of everything I just swallowed — will largely resist digestion, acting as dietary fiber and potential prebiotics, feeding specific populations of bacteria in my large intestine.
Because this is an espresso — unfiltered, prepared under high pressure — it contains meaningful amounts of cafestol and kahweol. My liver will process these diterpenes, and they will modestly influence my cholesterol metabolism.
The trigonelline that survived roasting is providing me with a dose of niacin. The Maillard reaction products are contributing antioxidant activity. The volatile aromatics — hundreds of them — are triggering olfactory receptors in my nasal cavity, creating the subjective experience of pleasure that keeps me coming back.
All of this is happening at once. All of it interacting. All of it modulated by my individual genetics, my habitual consumption, the state of my gut microbiome, what I ate for breakfast, and whether I’m sitting calmly at a café table or rushing to catch a train.
Every sip is a complex pharmacological event.
The rest of this book will show you exactly what’s happening, one computational lens at a time. We’ll start, in the next chapter, with the raw materials — the chemical fingerprint of the green coffee bean, mapped with the precision of modern mass spectrometry. Then we’ll follow those molecules through roasting, through extraction, through digestion, through metabolism, and into the tissues where they exert their effects.
By the end, you’ll never look at your morning cup the same way again.
Next: Chapter 2 — “The Green Bean’s Chemical Fingerprint”